Use data from Table 4C.1 to calculate the vapor pressure of mercury at 275 K. TABLE 4C.1
Question:
Use data from Table 4C.1 to calculate the vapor pressure of mercury at 275 K.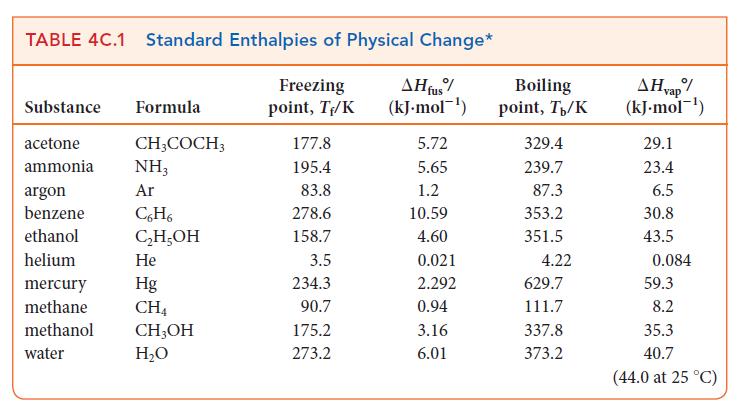
Transcribed Image Text:
TABLE 4C.1 Standard Enthalpies of Physical Change* Freezing AH fus% (kJ. mol) point, T/K Substance Formula acetone ammonia argon benzene ethanol helium CH3COCH3 NH3 Ar C6H6 CH5OH He Hg CH4 mercury methane methanol CHOH water HO 177.8 195.4 83.8 278.6 158.7 3.5 234.3 90.7 175.2 273.2 5.72 5.65 1.2 10.59 4.60 0.021 2.292 0.94 3.16 6.01 Boiling point, T/K 329.4 239.7 87.3 353.2 351.5 4.22 629.7 111.7 337.8 373.2 AH vap/ (kJ.mol-) 29.1 23.4 6.5 30.8 43.5 0.084 59.3 8.2 35.3 40.7 (44.0 at 25 C)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Table 4C1 contains the enthalpy of vaporization and ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Use data from Table 4C.1 to calculate the vapor pressure of methanol at 25.0 C. TABLE 4C.1 Standard Enthalpies of Physical Change* Freezing AH fus% (kJ.mol-) point, T/K Substance Formula acetone...
-
You are a chemical engineer at a processing plant for soft drinks. You have been asked to determine whether a particular solution will have a significantly different vapor pressure from pure water....
-
Use data from Appendix D to estimate (a) The normal boiling point of mercury and (b) The vapor pressure of mercury at 25 C. TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1...
-
The following balance sheets were prepared for Pam Corporation and Slest Company on January 1, 20X2, just before they entered into a business combination: Pam acquired all of Slest Companys assets...
-
A binary ideal-gas mixture of A and B undergoes an isothermal, isobaric separation at To, the infinite surroundings temperature. Starting with Eq. (4), Table 2.1, derive an equation for the minimum...
-
One of the most controversial topics to affect the accounting profession has been that of earnings management. Companies have been trying to manage their earnings to match analysts projections. While...
-
A model for a hemodialyser with simulation of the patient-artificial-kidney system: a case-study problem. A useful case study is the paper by Ramachandran and Mashelkar (1980), where a mesoscopic...
-
Doublewide Dealers has an ROA of 10 percent, a 2 percent profit margin, and a return on equity equal to 15 percent. What is the companys total assets turnover? What is the firms equity multiplier?
-
On January 1, JKR Shop had $550,000 of beginning inventory at cost. In the first quarter of the year, it purchased $1,690,000 of merchandise, returned $24,100, and paid freight charges of $38,600 on...
-
Go to the Webinars worksheet. DeShawn wants to determine the number of webinars the company can hold on Tuesdays and Thursdays to make the highest weekly profit without interfering with...
-
Evaluate K c for each of the following equilibria from the value of K: (a) 2 SO(g) + O(g) 2 SO3(g), K = 3.4 at 1000. K (b) NHHS(s) - NH3(g) + HS(g), K = 9.4 x 10- at 24 C
-
In a gas-phase equilibrium mixture of SbCl 5 , SbCl 3 , and Cl 2 at 500. K, PSbCl 5 = 0.072 bar and PSbCl 3 = 5.02 mbar. Calculate the equilibrium partial pressure of Cl 2 , given that K = 3.5 * 10 4...
-
Prove that if D is an integral domain, then D [ x] is an integral domain.
-
A return distribution of skilled managers that is highly distinct from the return distribution of unskilled managers, most likely implies a: A. highly efficient market. B. low opportunity cost of not...
-
The manager selection process begins by defining the universe of feasible managers. When defining this manager universe, the selection process should avoid: A. excluding managers based on historical...
-
A manager has a mandate to be fully invested with a benchmark that is a blend of largecap stocks and investment-grade bonds. Which of the following is not an indication that style drift has occurred?...
-
Which of the following statements is consistent with the manager adhering to a stated investment philosophy and investment decision-making process? A. Senior investment team members have left to form...
-
Which of the following investment vehicles provide investors with the highest degree of liquidity? A. Open-end funds B. Private equity funds C. Limited partnerships
-
What is the present value of $1,000, received ten years from now, using a periodic interest rate of 12% compounded annually?
-
The cash records of Holly Company show the following four situations. 1. The June 30 bank reconciliation indicated that deposits in transit total $720. During July, the general ledger account Cash...
-
Consider all of the different C!H bonds in cyclopentene, and rank them in order of increasing bond strength: H.
-
Explain the significance of the Boyle temperature.
-
Rank each group of radicals in order of increasing stability: (a) (b)
-
(a) Find an equation of the line that passes through the point (0, 9) and is parallel to 5x + 3y = 4. I
-
Common stock value-Constant growth Personal Finance Problem Telephone has paid the dividends shown in the following table. expected to be $5.06. Over the past 6 years, Elk County The firm's dividend...
-
Solve the equation and check your solution. 6[4y 2(y-1)] 2 + 7y = 0

Study smarter with the SolutionInn App


