Use data from Table 4C.1 to calculate the vapor pressure of methanol at 25.0 C. TABLE 4C.1
Question:
Use data from Table 4C.1 to calculate the vapor pressure of methanol at 25.0 °C.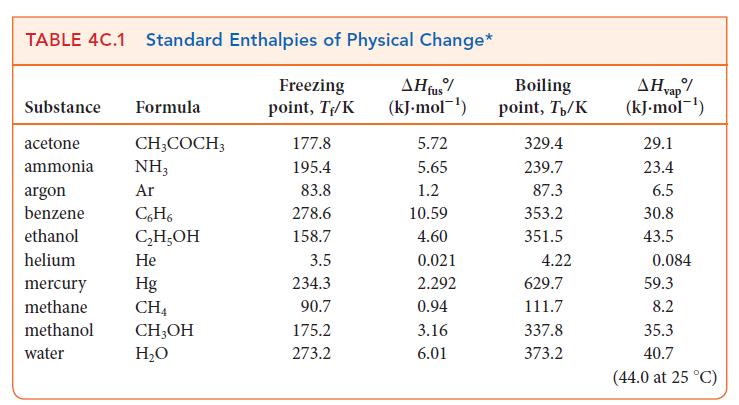
Transcribed Image Text:
TABLE 4C.1 Standard Enthalpies of Physical Change* Freezing AH fus% (kJ.mol-¹) point, T₁/K Substance Formula acetone ammonia argon benzene ethanol helium CH3COCH3 NH3 Ar C6H6 C₂H₂OH He Hg CH4 mercury methane methanol CH₂OH water H₂O 177.8 195.4 83.8 278.6 158.7 3.5 234.3 90.7 175.2 273.2 5.72 5.65 1.2 10.59 4.60 0.021 2.292 0.94 3.16 6.01 Boiling point, T/K 329.4 239.7 87.3 353.2 351.5 4.22 629.7 111.7 337.8 373.2 AH vap/ (kJ.mol-¹) 29.1 23.4 6.5 30.8 43.5 0.084 59.3 8.2 35.3 40.7 (44.0 at 25 °C)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
P 250 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
You are a chemical engineer at a processing plant for soft drinks. You have been asked to determine whether a particular solution will have a significantly different vapor pressure from pure water....
-
Calculate the vapor pressure at 25 C of a solution containing 99.5 g sucrose (C 12 H 22 O 11 ) and 300.0 mL water. The vapor pressure of pure water at 25 C is 23.8 torr. Assume the density of water...
-
Use data from Table 4C.1 to calculate the vapor pressure of mercury at 275 K. TABLE 4C.1 Standard Enthalpies of Physical Change* Freezing AH fus% (kJ. mol) point, T/K Substance Formula acetone...
-
Tell whether the given side lengths of a triangle can represent a right triangle. 36, 48, and 60
-
Describe three methods suitable for the separation of air into nitrogen and oxygen.
-
How can a firm speed up its awareness of emerging problems so that it can take corrective action? Be as specific as you can.
-
Although radiation is important in heat transfer, an analogous model can be used in the design of photochemical reactors. The modeling of these reactors requires that the radiation intensity be...
-
A diet center claims that it has the most effective weight loss program in the region. Its advertisements say, Participants in our program lose more than 5 pounds within a month. Six clients of this...
-
Crown Brewing issued new shares in 2021 but some investors had not paid their capital\ contribution by December 31st, 2021. Hence, Crown Brewing recognizes a contributed capital\ receivable. No other...
-
Using exponential smoothing, calculate the forecasts for the same months as in problem 8.3c. The old average for month 3 was 96 and = 0.4. What is the difference between the two forecasts for month...
-
The density of a 5.00% by mass K 3 PO 4 aqueous solution is 1.043 g cm 3 . Determine (a) The molality; (b) The molarity of potassium phosphate in the solution.
-
Most reactions proceed faster at higher temperatures, and many industrial processes are carried out at high temperatures. However, for exothermic reactions increasing the temperature reduces the...
-
Wamser Companys standard labor cost per unit of output is $20 (2 hours _ $10 per hour). During August, the company incurs 2,100 hours of direct labor at an hourly cost of $10.50 per hour in making...
-
Which of Yellows statements regarding the trade implementation of non-equity investments is correct? A. Only Statement 4 B. Only Statement 5 C. Both Statement 4 and Statement 5 Robert Harding is a...
-
Identify which investment manager is most suitable for Patnode. Justify your response based solely on each managers investment philosophy and approach Susan Patnode, age 66, was recently widowed and...
-
Discuss how Smiths stated expectation would be reflected in estimated portfolio risk under the fee structure identified by Porter. After narrowing their choice to three managers with different fee...
-
Determine which of the three investment vehicles is most appropriate for Circues IPS. Justify your response. Porter and Smith next consider how the performance-based fee structures of the prospective...
-
An investor is considering hiring three managers who have the following skill levels: Type I and Type II errors both occur when the investor is: A. hiring Manager 1 for large-cap stocks and not...
-
Your company paid employees who were eligible for work opportunity credit $25,000 last year. Of these wages, $21,000 is eligible for a tax credit of 40% of the wages. The remaining wages are eligible...
-
This problem continues the Draper Consulting, Inc., situation from Problem 12-45 of Chapter 12. In October, Draper has the following transactions related to its common shares: Oct 1 Draper...
-
Using 2-propanol as your only source of carbon, show how you would prepare 2-methyl-2-pentanol.
-
A bottle at 325 K contains an ideal gas at a pressure of 162.5 10 3 Pa. The rubber stopper closing the bottle is removed. The gas expands adiabatically against P external = 120.0 10 3 Pa, and some...
-
Predict the product and draw the mechanism for each of the following reactions: a. b. c. 1) LAH 2) H20 1) LAH 2) H20
-
State the conditions under which the Moving Average method can be recommended for trend analysis. How will you determine the period of the moving average? (a) Calculate the 4-year moving average of...
-
Consider the model of insurance markets discussed in class. Suppose the consumer's expected utility function is U(1, 2) = (1 - p) Inx + pln2, where p is the probability of loss, is consumption in the...
-
42L T 6. Given that V - Use partial derivative to approximate the K2 maximum percentage error in estimating V, if the error in estimating K, if the error in measuring L and V are 4% and 1%. [6 marks]...

Study smarter with the SolutionInn App


