Write a balanced chemical equation for the formation reaction of (a) HCl(g); (b) C 6 H 6
Question:
Write a balanced chemical equation for the formation reaction of
(a) HCl(g);
(b) C6H6(l);
(c) CuSO4 · 5H2O(s);
(d) CaCO3(s, calcite). For each reaction, determine ΔH°, ΔS°, and ΔG° from data in Appendix 2A.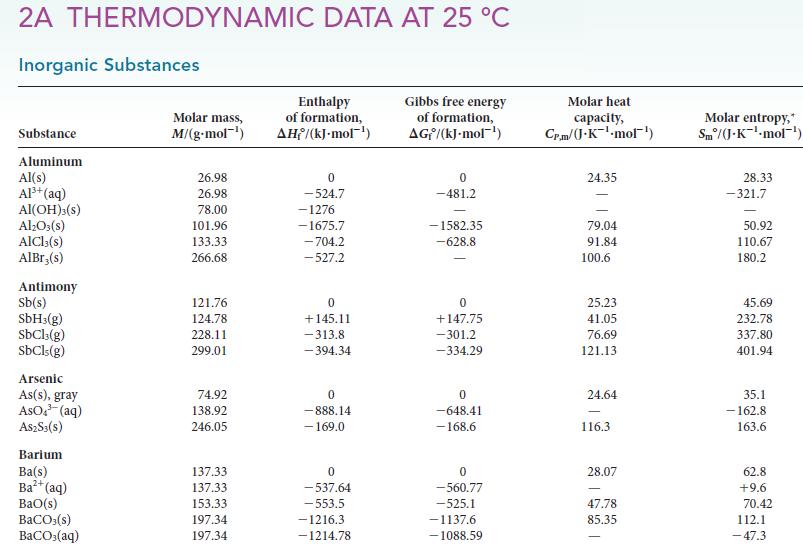
Transcribed Image Text:
2A THERMODYNAMIC DATA AT 25 °C Inorganic Substances Substance Aluminum Al(s) Al³+ (aq) Al(OH)3(S) Al₂O3(s) AlCl3(s) AlBr,(s) Antimony Sb(s) SbH3(g) SbCl3(g) SbCls (g) Arsenic As(s), gray AsO³(aq) A$2S3(S) Barium Ba(s) Ba²+ (aq) BaO(s) BaCO3(s) BaCO3(aq) Molar mass, M/(g.mol-¹) 26.98 26.98 78.00 101.96 133.33 266.68 121.76 124.78 228.11 299.01 74.92 138.92 246.05 137.33 137.33 153.33 197.34 197.34 Enthalpy of formation, AH/(kJ-mol-¹) 0 -524.7 -1276 -1675.7 -704.2 -527.2 0 +145.11 -313.8 -394.34 0 -888.14 - 169.0 0 -537.64 -553.5 -1216.3 -1214.78 Gibbs free energy of formation, AG/(kJ.mol-¹) 0 -481.2 -1582.35 -628.8 0 +147.75 -301.2 -334.29 0 -648.41 -168.6 0 -560.77 -525.1 -1137.6 -1088.59 Molar heat capacity, Cr.m/(J.K¹.mol¹) 24.35 79.04 91.84 100.6 25.23 41.05 76.69 121.13 24.64 116.3 28.07 47.78 85.35 Molar entropy,* Sm/(J-K-¹-mol¹) 28.33 -321.7 50.92 110.67 180.2 45.69 232.78 337.80 401.94 35.1 -162.8 163.6 62.8 +9.6 70.42 112.1 -47.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Lets write the balanced chemical equations for the formation reactions of the compounds youve mentioned and determine their standard enthalpy H entrop...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Write a balanced chemical equation for the formation reaction of (a) NH 3 (g); (b) H 2 O(g); (c) CO(g); (d) NO 2 (g). For each reaction, determine H, S, and G from data in Appendix 2A. 2A...
-
(a) Write a balanced chemical equation for the formation of 1 mol of MgO(s) from the elements in their standard states. (Find the value for f H for MgO(s) in Appendix L.) (b) What is the standard...
-
Write a balanced chemical equation for each neutralization reaction in Exercise 3. In exercise 5 a. HCl and KOH b. H2SO4 and KOH c. H3PO4 and Ni(OH)2
-
Wilburton Hospital is investigating the possibility of investing in new dialysis equipment. Two local manufacturers of this equipment are being considered as sources of the equipment. After-tax cash...
-
The cost of merchandise sold for Kohl's Corporation for a recent year was $9,891 million. The balance sheet showed the following current account balances (in millions): Determine the amount of cash...
-
Find the points of intersection of the pairs of curves. y = 1/2x 3 - 2x 2 , y = 2x
-
Darwin Ltd wants to focus on people, profits, planet. The board of directors has proposed linking top managers pay to broad measures of environmental sustainability, and worker and customer...
-
Imagine you are an HR executive at P&G. Given the company's strategic orientation toward purpose and values, what would you do to help align a new performance management system with the strategic...
-
The definition, " ' Chiropractor ' means a person who is a medical quack with no legitimate scientific background," is an example of: Theoretical definition Definition by subclass Persuasive...
-
Suppose that 320.0 g of ethanol at 18.0C is mixed with 120.0 g of ethanol at 56.0 C at constant atmospheric pressure in a thermally insulated vessel. Calculate S and S tot for the process.
-
(a) Calculate the work associated with the isothermal, reversible expansion of 1.000 mol of ideal gas molecules from 7.00 L to 15.50 L at 25.0 C. (b)Calculate the work associated with the...
-
Use the identity to prove that Then use the inequality Finally, prove rigorously that y y sin (+1) cos (**) 2 sin x + sin y = 2 sin
-
Lady MacBath is a business selling bottles of dry cleaning solvent (spot remover) for \(\$ 10\) each. The variable cost for each bottle is \(\$ 4\). Lady MacBath's total fixed cost for the year is...
-
Suppose you work for the Miniola Hills Bus Company. The business' ten buses made a total of 80 trips per day on 310 days last year, for a total of 350000 kilometres. Another year like last year will...
-
The sales budget for Astra Awards Company shows budgeted sales (in awards) for December and the first four months of next year: Required: Prepare a budget for the number of awards Astra needs to...
-
The sales budget for Merita Medallion Company shows budgeted sales (in medallions) for December and the first four months of next year: Required: Prepare a budget for the number of medallions Merita...
-
Well Feed Company sells pet food in 10 -kilogram bags for \(\$ 12.40\) per bag. Sales estimates for the first three months of the year are as follows: December sales were 16000 bags of pet food. Well...
-
a. Identify the equilibrium outcome(s) in each of the three payoff tables. b. In each table, predict the exact outcome that will occur and explain your reasoning. c. In Table III, suppose the column...
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
At a certain temperature, the vapor pressure of pure benzene (C 6 H 6 ) is 0.930 atm. A solution was prepared by dissolving 10.0 g of a nondissociating, nonvolatile solute in 78.11 g of benzene at...
-
The vapor pressure of a solution containing 53.6 g glycerin (C 3 H 8 O 3 ) in 133.7 g ethanol (C 2 H 5 OH) is 113 torr at 40C. Calculate the vapor pressure of pure ethanol at 40C assuming that...
-
In terms of Raoults law, distinguish between an ideal liquidliquid solution and a nonideal liquidliquid solution. If a solution is ideal, what is true about H soln , T for the solution formation, and...
-
debt valued at $20 billion on its balance sheet, while the market value of its common stock is roughly $40 billion. The yield to maturity on the debt is 5.50%, the cost of equity for the firm is 12%,...
-
Mardaloop Inc. is developing a new process which it plans to sell. During 2016, 2017, the company had capitalized $1.5 million and $0.3 million respectively. An additional $0.4 million was spent in...
-
You are the owner of the local record store, and you are considering opening a coffee shop in a vacant area in the back of the store. You estimate that it will cost you $50,000 to set up the store...

Study smarter with the SolutionInn App


