Write a balanced chemical equation for the formation reaction of (a) NH 3 (g); (b) H 2
Question:
Write a balanced chemical equation for the formation reaction of
(a) NH3(g);
(b) H2O(g);
(c) CO(g);
(d) NO2(g). For each reaction, determine ΔH°, ΔS°, and ΔG° from data in Appendix 2A.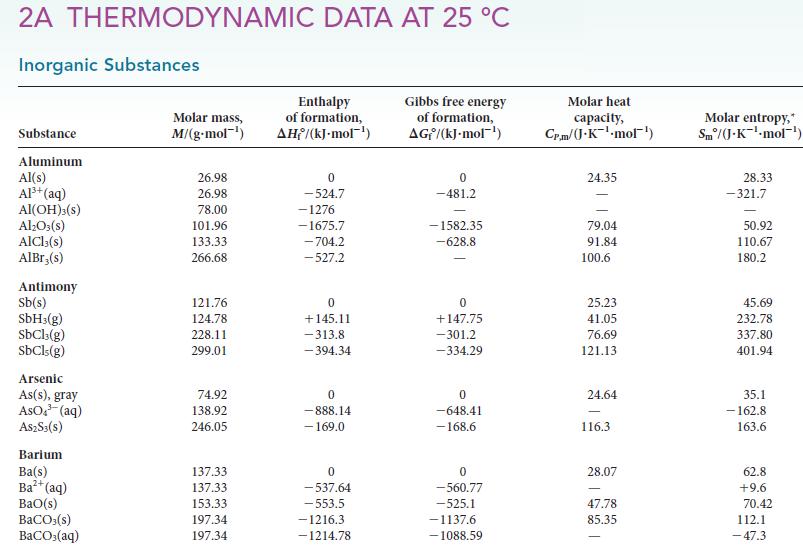
Transcribed Image Text:
2A THERMODYNAMIC DATA AT 25 °C Inorganic Substances Substance Aluminum Al(s) Al³+ (aq) Al(OH)3(S) Al₂O3(s) AlCl3(s) AlBr,(s) Antimony Sb(s) SbH3(g) SbCl3(g) SbCls (g) Arsenic As(s), gray AsO³(aq) A$2S3(S) Barium Ba(s) Ba²+ (aq) BaO(s) BaCO3(s) BaCO3(aq) Molar mass, M/(g.mol-¹) 26.98 26.98 78.00 101.96 133.33 266.68 121.76 124.78 228.11 299.01 74.92 138.92 246.05 137.33 137.33 153.33 197.34 197.34 Enthalpy of formation, AH/(kJ-mol-¹) 0 -524.7 -1276 -1675.7 -704.2 -527.2 0 +145.11 -313.8 -394.34 0 -888.14 - 169.0 0 -537.64 -553.5 -1216.3 -1214.78 Gibbs free energy of formation, AG/(kJ.mol-¹) 0 -481.2 -1582.35 -628.8 0 +147.75 -301.2 -334.29 0 -648.41 -168.6 0 -560.77 -525.1 -1137.6 -1088.59 Molar heat capacity, Cr.m/(J.K¹-mol¹) 24.35 79.04 91.84 100.6 25.23 41.05 76.69 121.13 24.64 116.3 28.07 47.78 85.35 Molar entropy,* Sm/(J.K¹-mol-¹) 28.33 -321.7 50.92 110.67 180.2 45.69 232.78 337.80 401.94 35.1 -162.8 163.6 62.8 +9.6 70.42 112.1 -47.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Ng Hg NH3g AH 4611 kJmol AS 9938 JKmol AG 1...View the full answer

Answered By

Ashish Jaiswal
I have completed B.Sc in mathematics and Master in Computer Science.
4.90+
20+ Reviews
39+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Write a balanced chemical equation for the formation reaction of (a) HCl(g); (b) C 6 H 6 (l); (c) CuSO 4 5H 2 O(s); (d) CaCO 3 (s, calcite). For each reaction, determine H, S, and G from data in...
-
(a) Write a balanced chemical equation for the formation of 1 mol of MgO(s) from the elements in their standard states. (Find the value for f H for MgO(s) in Appendix L.) (b) What is the standard...
-
Write a balanced chemical equation for the pentose phosphate pathway in the first two modes depicted in Figure 12.36, where (a) ribose-5-phosphate synthesis is maximized (b) NADPH production is...
-
If you sign up for a savings account with an annual interest rate of 12% (1% per month) that you will receive 4 million won in 2 years, what is the monthly amount you have to pay? However, suppose...
-
A simple capacitor is a device formed by two insulated conductors adjacent to each other. If equal and opposite charges are placed on the conductors, there will be a certain difference of potential...
-
When a cars brakes are slammed on at a speed of x miles per hour, the stopping distance is 1/20x 2 feet. Show that when the speed is doubled the stopping distance increases fourfold.
-
Explain why composition of the Board of Directors is important.
-
The following selected accounts were taken from the financial records of Santa Rosa Distributors at December 31, 2016. All accounts have normal balances. Cash ................. $ 26,760 Accounts...
-
What piece of hardware connects a peripheral device like a keyboard or mouse to the system bus? 1 point a device driver a device controller a semaphore a network port
-
Calculate the standard reaction entropy, enthalpy, and Gibbs free energy for each of the following reactions from data in Appendix 2A: (a) The decomposition of hydrogen peroxide: (b) The preparation...
-
Initially an ideal gas at 323 K occupies 1.67 L at 4.95 atm. The gas is allowed to expand to 7.33 L by two pathways: (a) Isothermal, reversible expansion; (b) Isothermal, irreversible free expansion....
-
John, Lesa, and Tabir form a limited liability company. John contributes 60 percent of the capital, and Lesa and Tabir each contribute 20 percent. Nothing is decided about how profits will be...
-
Suppose that firms were expecting inflation to be 3 percent, but it actually turned out to be 7 percent. Other things equal, firm profits will be: a. smaller than expected. b. larger than expected.
-
List five continuous monitoring/auditing routines you would recommend to a client for a payment processing operation. Specify what data would be collected, how it would be analyzed, the reporting...
-
What are the implications of Kurt Lewins Step 3: Refreeze for operational auditors and how can they verify that management has effectively implemented this step?
-
Provide three examples of how organizations can create continuous process flows to bring problems to the surface (principle 2).
-
Suppose that firms are expecting 6 percent inflation while workers are expecting 9 percent inflation. How much of a pay raise will workers demand if their goal is to maintain the purchasing power of...
-
In the following game tree, players A and B alternate moves. At each turn, a player can terminate the game or pass the move to the next player. By passing, the player increases the rivals potential...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
Name each of the following alkenes and alkynes. a. b. c. d. e. f. CH3 CH3CH-C=CH
-
Name each of the following organic compounds a. b. c. d. e. CH3CHCHCHCHCHCH
-
Element 106 has been named seaborgium, Sg, in honor of Glenn Seaborg, discoverer of the first transuranium element. a. Write the expected electron configuration for element 106. b. What other element...
-
Jake has $44,000 invested in Vanguard Money Market Mutual Fund for 71 days at a 7-day yield of 3.62%. How much interest will he receive in 71 days?
-
A new father plans on saving for his daughter's college education. He will donate $1,000 on her first birthday. After that, he will increase his donation by 4.50% each year and will make his last...
-
With interest rates near an all-time low, a family decides to purchase their dream home. The house will cost $350,000. The family will pay 20% as a down payment, and finance the remaining balance...

Study smarter with the SolutionInn App


