A gas mixture is composed of (15 %) carbon monoxide, (10 %) methane, and the rest air.
Question:
A gas mixture is composed of \(15 \%\) carbon monoxide, \(10 \%\) methane, and the rest air. Is this mixture flammable? Use the flammability data provided in Appendix B.
Data From Appendix B:
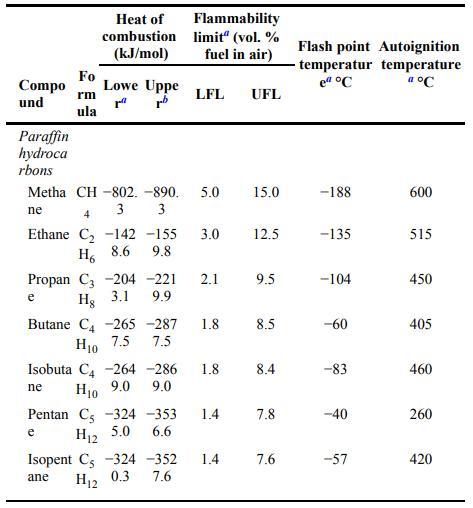
Transcribed Image Text:
Compo und Paraffin hydroca rbons Fo rm ula Heat of combustion (kJ/mol) Lowe Uppe rrb Metha CH-802. -890. 5.0 ne 3 3 e Flammability limit" (vol. % fuel in air) 4 Ethane C -142 -155 3.0 9.8 H 8.6 LFL Propan C3 204 -221 2.1 9.9 Hg 3.1 e Butane C4 -265-287 7.5 H10 7.5 Pentan C -324 -353 6.6 H12 5.0 Isopent C -324 -352 7.6 ane H12 0.3 1.8 1.4 UFL Isobuta C4 -264 -286 1.8 8.4 Hio 9.0 9.0 ne 1.4 15.0 12.5 9.5 8.5 7.8 7.6 Flash point Autoignition temperatur temperature el C a C -188 -135 -104 -60 -83 -40 -57 600 515 450 405 460 260 420
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The gas mixture is composed of 15 15 15 carbon monoxide CO and 1...View the full answer

Answered By

Divishad Londhe
I have completed my undergraduate studies as as mechanical engineer and my research focus was Robotics. I have also given GRE and can teach people about prep. I have conducted many robotics workshops in high schools and colleges and taught almost 100-200 students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
Draw an approximate flammability triangle diagram for methyl alcohol. Use published flammability data from Appendix B and Table 6-3. If a gas containing 20\% methyl alcohol, \(5 \%\) oxygen, and \(75...
-
A gas mixture is found to contain two diatomic A and B species for which the partial pressures of both are 0.05065 MPa (0.5 atm). This mixture is to be enriched in the partial pressure of the A...
-
The financial statements for Leon's are provided in Appendix B at the end of the text. Determine the free cash flow for the year ended December 31, 2011. The $24,999,000 purchase of property, plant,...
-
A car is initially travelling at 3 0 m / s , which is above the speed limit . The driver sees a speed limit trap ahead and applies the brakes for 5 seconds, causing the car to slow down by 2 m / s...
-
Determine the volume and the surface area of the solid obtained by rotating the area of Prob. 5.2 about (a) The x axis, (b) The lines 19 x = in. 9 in 10 in.- 12 In. Sin.
-
You are planning to build a new home with approximately 2,0002,500 gross square feet of living space on one floor. In addition, you are planning an attached two-car garage (with storage space) of...
-
On March 13, 1963, Ernesto Miranda was arrested for kidnapping and rape and taken to a Phoenix police station. After being identified by the complaining witness, he was questioned by two police...
-
Zebra Imaginarium, a retail business, had the following cash receipts during December 20--. The sales tax is 6%. Dec. 1 Received payment on account from Michael Anderson, $1,360. 2 Received payment...
-
2. Draw one (1) accurate graph to show the production possibilities frontier for the head of household's own child PPFo given a budget of T=20Eo+20Co when E is units of education and C is units of...
-
Estimate the upper and lower flammable limits for carbon monoxide and heptane using the stoichiometric method via Equations 6-10 and 6-11 in the text. Compare to experimental values provided in...
-
What is the flash point temperature \(\left(ight.\) in \(\left.{ }^{\circ} \mathrm{C}ight)\) of a 50 mole percent mixture of methanol and water? Use Appendix B for the flash point temperature for...
-
A given mass of an ideal gas occupies a volume of 4.00 m 3 at 758 mmHg. Compute its volume at 635 mmHg if the temperature remains unchanged.
-
Let's consider further the role that benefits play in recruiting. Put yourself in the role of your organization's recruiter, what benefits might you use to attract the generation of workers (under 45...
-
How long would it take an investment to double under each of the following conditions? (a) Interest is 4.2% compounded monthly. (Enter your answer as a whole number of years and months. Round your...
-
QUESTION Explain the following *multi-professional *Interprofessional working *Ethics and its role in interprofessional practise *Roles and responsibility of each healthcare team member...
-
An 80-percent-efficient pump with a power input of 36.00 hp is pumping water from a lake to a nearby pool at a rate of 1.5 ft /s through a constant-diameter pipe. The free surface of the pool is 80...
-
1. As mentioned in the case Starbuck limits the team size usually from three to six employees, why is the size of team an important factor? What are the benefits to Starbucks of having small teams of...
-
Journalize the following transactions using the allowance method of accounting for uncollectible receivables: July 7. Received $500 from Betty Williams and wrote off the remainder owed of $2,000 as...
-
Chao, Louis, and Mari, unrelated individuals, own all of the shares of Cerise Corporation. All three shareholders have been active in the management of Cerise since its inception. In the current...
-
Some scientists believe that the collision of one or more large asteroids with the Earth was responsible for the extinction of the dinosaurs. The unit of kiloton is used to describe the energy...
-
A structure known as a cantilever beam is clamped at one end but free at the other, analogous to a diving board that supports a swimmer standing on it (Figure P3.28). Using the following procedure,...
-
Perform measurements as described in Problem P3.28 for cantilever beams of several different lengths. Can you show experimentally that, for a given force F, the deflection of the cantilevers tip is...
-
7. If mZLMP is 11 degrees more than mZNMP and mZNML = 137, find each measure. L M P N m/LMP = mZNMP =
-
Why is a marketing strategy crucial for a business? What is it?
-
Part A Discuss the benefits of having a strong and active Diversity Equity and Inclusion Champion in the firm. Part B What are the challenges faced by women in the modern workplace when it comes on...

Study smarter with the SolutionInn App


