A 0.229-g sample of an unknown monoprotic acid is titrated with 0.112 M NaOH. The resulting titration
Question:
A 0.229-g sample of an unknown monoprotic acid is titrated with 0.112 M NaOH. The resulting titration curve is shown here.
Determine the molar mass and pKa of the acid.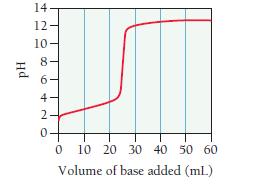
Transcribed Image Text:
Hd 14 12- 10 8 6- Na 00 4- 0 0 10 20 30 40 50 60 Volume of base added (ml)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
pK...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A 0.446-g sample of an unknown monoprotic acid is titrated with 0.105 M KOH. The resulting titration curve is shown here. Determine the molar mass and pKa of the acid. Hd 14 12- 10- 06+NO 8- 4- 2 20...
-
Find the analytic tan te=4+Pv. 37 co20-9 Coso +2.
-
A 0.1276-g sample of an unknown monoprotic acid was dissolved in 25.0 mL of water and titrated with 0.0633 M NaOH solution. The volume of base required to bring the solution to the equivalence point...
-
The following question is designed to highlight key concepts from the Loyalty Programs topic article titled, "StarBUCKS, Loyalty, and Breakage" (Nevraumont 2019). Q. Author's position. "If you hire...
-
Peters Company produces golf discs which it normally sells to retailers for $7 each. The cost of manufacturing 20,000 golf discs is: Materials ........ $ 10,000 Labor ......... 30,000 Variable...
-
During a particular year, the T-bill rate was 6%, the market return was 14%, and a portfolio manager with beta of .5 realized a return of 10%. a. Evaluate the manager based on the portfolio alpha. b....
-
How to draft a statement of fact for complaints?
-
You should recognize that basing a decision solely on expected returns is appropriate only for risk-neutral individuals. Because your client, like virtually everyone, is risk averse, the riskiness of...
-
Write SHORT CLASSIC ESSAY about the two articles below: Rice, W. (2020). Letter to a young Indigenous journalist. The Walrus. ...
-
A 20.0-mL sample of 0.115 M sulfurous acid (H 2 SO 3 ) solution is titrated with 0.1014 M KOH. At what added volume of base solution does each equivalence point occur?
-
A 25.0-mL sample of 0.125 M pyridine is titrated with 0.100 M HCl. Calculate the pH at each volume of added acid: 0 mL, 10 mL, 20 mL, equivalence point, one-half equivalence point, 40 mL, 50 mL....
-
How much gross income is recognized by Carol, who received $ 10,000 damages (two months salary) for pain and suffering because of the school administrations critical reaction to her negative comments...
-
What is the MD5 hash value ofmission.docx? What is required to be able to successfully recover a deleted file intact within unallocated space? Why is it important to extract embedded files within...
-
Company Culture and Competitive Analysis: introduce the corporate culture and competitive analysis of Mario's Place Coffee. Who are the key stakeholders and their roles in the company. Identify the...
-
Appraise the long-term solvency ratios as given in the table over the last three years in words. The presentation of your answer should be well-organized, impactful. The long-term solvency ratios are...
-
2. How many blocks are needed for the 10th staircase? 2 3
-
Identify at least two plausible service departments in your industry* (not including HR, IT or maintenance). Describe the departments, the services they provide to the producing departments or to...
-
SAS No. 99 identifies six characteristics of journal entries. Discuss the nature of each.
-
If the jobs displayed in Table 18.24 are processed using the earliestdue-date rule, what would be the lateness of job C? TABLE 18.24 Processing Times and Due Dates for Five Jobs Job C D E...
-
A point source S is a perpendicular distance R away from the center of a circular hole of radius a in an opaque screen. If the distance from S to the periphery of the hole is (R + ), show that...
-
Starting with Eq. (9.53) for the transmitted wave, compute the flux density, that is, Eq. (9.54). , = Egelo tt' Eoeiot 1 pPe-i (9.53) 14(t')? I; (1 + r4) 2r (9.54) cos 8
-
A form of the Jamin Interferometer is illustrated in Fig. P.9.52. How does it work? To what use might it be put? Figure P.9.52
-
A car of mass m moving at a speed v 1 collides and couples with the back of a truck of mass 2 m moving initially in the same direction as the car at a lower speed v 2 . (a) What is the speed v f of...
-
A couple of wave turbines are 1 5 m apart in Galway Bay. They both rise and fall every 1 9 . 9 seconds ( period ) . When one turbine is at its highest point, the other turbine is at its lowest point....
-
A B Water (density = 1000 kg/m^3) flows in a horizontal circular pipe from a region of larger cross section A = 7.8x10^-3 m^2 to smaller cross section B = 2.3x10^-3 m^2 as shown. If the speed of the...

Study smarter with the SolutionInn App


