Balance each redox reaction occurring in acidic aqueous solution. a. I (aq) + NO (aq) b. CIO4
Question:
Balance each redox reaction occurring in acidic aqueous solution.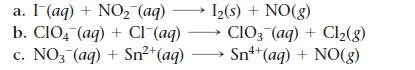
Transcribed Image Text:
a. I (aq) + NO₂ (aq) b. CIO4 (aq) + Cl¯(aq) c. NO3(aq) + Sn²+ (aq) 1₂(s) + NO(g) CIO3(aq) + Cl₂(g) Sn+ (aq) + NO(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a Iaq NOaq Is NOg Halfreactions Oxidation Iaq Is 2e Reduction NOaq Haq e NOg HOl Balance the oxidati...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
PART A: Balance each redox reaction occurring in acidic aqueous solution. MnO4( a q )+Al( s )Mn2+( a q )+Al3+( a q ). Express your answer as a chemical equation. Identify all of the phases in your...
-
Balance each redox reaction occurring in acidic aqueous solution. a. PbO(s) + I (aq) Pb+ (aq) + 1(s) 2+ b. SO32 (aq) + MnO4 (aq) SO42 (aq) + Mn+ (aq) 2- c. S03 (aq) + Cl(g) SO4 (aq) + Cl (aq)
-
Balance each redox reaction occurring in acidic aqueous solution. a. Zn(s) + Sn+ (aq) b. Mg(s) + Cr+ (aq) Zn+ (aq) + Sn(s) Mg+ (aq) + Cr(s) c. MnO4 (aq) + Al(s) Mn+ (aq) + A1+ (aq)
-
You require inventory and accounts receivable collateral for all C&I loans. You have a guideline of an advance rate of 70% for customer receivables of less than 60 days of age. Older receivables get...
-
Lago Corporation is considering adopting the standard costing method. Dan Sarkis, the manager of the Ohio Division, attended a corporate meeting at which Leah Rohr, the controller, discussed the...
-
1. Discuss why financial statement users find environmental disclosures informative. 2. Obtain the G3 sustainability reporting guidelines of the Global Reporting Initiative (GRI,...
-
For the following products and countries, identify the type of warehouse that should be used as well as the method of transportation that should deliver the product to end users. Using the Internet,...
-
You intend to use MUS as a part of the audit of several accounts for Roynpower Manufacturing Company. You have done the audit for the past several years, and there has rarely been an adjusting entry...
-
Deliverable length: 300 - 400 words for four discussions. Compare and contrast benefits and challenges that exist between centralized database management systems and distributed database management...
-
Balance each redox reaction occurring in basic aqueous solution. a. HO(aq) + ClO(aq) CIO (aq) + O(g) b. Al(s) + MnO4 (aq) MnO(s) + Al(OH)4 (aq) c. Cl(g) Cl(aq) + CIO (aq)
-
How can the corrosion of iron be prevented?
-
Using the following information, decide whether Marks Video Games should close any of itsstores: Clawson Store Redford Store Port Huron Store Sales revenue Variable costs Direct fixed costs Indirect...
-
company that is using its assets at only 85% of their full capacity generated Sales of $680,000 last year. The company's Net Profit Margin was 7.6% and they paid dividends equal to 22% of net profit....
-
In 2022, Rich (28) lived with his daughter, Ava (4), his brother, Joe (18), and his fiance, Linda (30) for the entire year. Rich's adjusted gross income is $36,555, Joe's gross income is $4,100,...
-
Display the American flag using a nested loop of these dimensions: 7x20 flag 4x7 stars as Alternate and to represent stripe colors ***** === ======
-
Connor and Martin are contemplating international business in their industry and feel that global expansion is a great transition for the company. They do not feel as if they understand IFRS and are...
-
In Fig. 6-14, a mass of 30 kg Is supported by a compression member CD and two tension members AC and BC. CD makes an angle of 400 with the wall. A, B, and Care in a horizontal .plane. AEEB-1000 mm....
-
The American Flag Corporation (AFC) wants to determine the effect of its inventory turnover and days sales outstanding (DSO) on its cash flow cycle. Last year, AFC's sales (all on credit) were...
-
The percentage of completion and completed contract methods are described in the FASB ASC. Search the codification to find the paragraphs covering these topics, cite them, and copy the results.
-
Estimate the pressure exerted by your feet when you stand upright on the floor. Compare it to the pressure when you are wearing stiletto heels (use the value at the heel).
-
When traveling in a commercial airplane, you are sometimes given snacks, such as peanuts, in a sealed bag. The bag is often bulging much more than when you purchase a bag of peanuts in a store....
-
When a typical 1200-page textbook is sitting on a table, it exerts a force on the table. Estimate the associated pressure.
-
Liz's Volleyball Warehouse offers credit terms of 2/10, n/30. The local taxing authority requires a 5% tax on sales to final consumers. On July 11, The Volleyball Academy purchased equipment costing...
-
Watson's fiscal year end is December 31. In July 2019, the company had purchased 80,000 shares of Holmes Corporation common stock for $15.75 per share as an equity investment. At the end of 2021, the...
-
Mohammad has worked for Almond Tire's for 7 years. His vacation pay is included in his pay each week. Since he has been with this company more than 5 years he now receives 6% vacation pay. He makes...

Study smarter with the SolutionInn App


