Balance each redox reaction occurring in acidic aqueous solution. a. PbO(s) + I (aq) Pb+ (aq)
Question:
Balance each redox reaction occurring in acidic aqueous solution.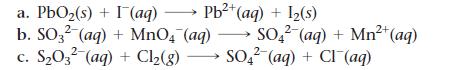
Transcribed Image Text:
a. PbO₂(s) + I (aq) →→→ Pb²+ (aq) + 1₂(s) 2+ b. SO32 (aq) + MnO4 (aq) → SO42² (aq) + Mn²+ (aq) 2- c. S₂03² (aq) + Cl₂(g) → SO4² (aq) + Cl¯ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a PbO s 21 aq 4H aq 2 Pb aq I2...View the full answer

Answered By

PU Student
cost accounting
financial accounting
auditing
internal control
business analyst
tax
i have 3 years experience in field of management & auditing in different multinational firms. i also have 16 months experience as an accountant in different international firms. secondary school certification.
higher secondary school certification.
bachelors in mathematics.
cost & management accountant
4.80+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
PART A: Balance each redox reaction occurring in acidic aqueous solution. MnO4( a q )+Al( s )Mn2+( a q )+Al3+( a q ). Express your answer as a chemical equation. Identify all of the phases in your...
-
Balance each redox reaction occurring in acidic aqueous solution. a. I (aq) + NO (aq) b. CIO4 (aq) + Cl(aq) c. NO3(aq) + Sn+ (aq) 1(s) + NO(g) CIO3(aq) + Cl(g) Sn+ (aq) + NO(g)
-
Balance each redox reaction occurring in acidic aqueous solution. a. Zn(s) + Sn+ (aq) b. Mg(s) + Cr+ (aq) Zn+ (aq) + Sn(s) Mg+ (aq) + Cr(s) c. MnO4 (aq) + Al(s) Mn+ (aq) + A1+ (aq)
-
What is the type of the expressions computed on these two lines? 4 > 5 print (4>5)
-
Lightning Industries specializes in making Flash, a high-moisture, low-alkaline wax used to protect and preserve skis. The company began producing a new, improved brand of Flash on January 1....
-
What is a gearing adjustment, and on what ideas is it based?
-
Choose a country from three of the regions presented in Table 6.7. Using the Internet, collect as much information as you believe is needed to identify the potential for market segments based on age,...
-
The scal year of Duchess County ends on December 31. Property taxes are due March 31 of the year in which they are levied. 1. Prepare journal entries (excluding budgetary and closing entries) to...
-
Assume the company is transitioning from a traditional file environment to a database management system / relational database. - Describe and explain the challenges / issues in transitioning from a...
-
Balance each redox reaction occurring in basic aqueous solution. a. HO(aq) + ClO(aq) CIO (aq) + O(g) b. Al(s) + MnO4 (aq) MnO(s) + Al(OH)4 (aq) c. Cl(g) Cl(aq) + CIO (aq)
-
How can the corrosion of iron be prevented?
-
Write a computer program that uses Equations (3.4) and the appropriate transformed data to estimate the parameters of the following models. a. y = bx n b. y = be ax c. y = a lnx + b d. y = ax 2 e. y...
-
Find an equation of the horizontal line through (1,5). The equation is
-
What does a quality recruit look like for a police officer position? What are the specific things that stand out to you as being important traits for an officer? Explain your reasoning for each...
-
2. Complete the following questions related to Bracket methods: (a) Write a function to perform the Bisection method for any scalar nonlinear equation f(x) = 0. The inputs to your function should be:...
-
If the Costa Rican government sets the exchange rate at 10 colon per U.S. dollar, then the supply of Costa Rican colones will increase. the supply of Costa Rican colones will decrease. the quantity...
-
(a) Explain what the price slippage is in the AMMS mechanism in DeFi. Do we have the price slippage in the order book mechanism? Show your reasons. (b) What are the needed conditions for an attacker...
-
Does the executive summary describe the key elements needed in a business plan? Is the proposed offer well written, why or why not? What are the important marketing issues and competitive advantages?...
-
Keating & Partners is a law firm specializing in labour relations and employee-related work. It employs 25 professionals (5 partners and 20 managers) who work directly with its clients. The average...
-
Figure Q10.16 shows a popular demonstration involving a moving fluid. Here an air jet is aimed upward and levitates a small object such as a table-tennis ball. The ball is drawn to the center of the...
-
The recommended pressure in your cars tires is usually specified as a gauge pressure. If a tire has a gauge pressure of 15 lb/in. 2 , what is the absolute pressure in pascals?
-
Explain how there can be dust on a moving fan blade.
-
Jamie wanted to establish an education fund for his 6-year old son, Michael. Jamie decided to transfer his shares in a local manufacturing company into an inter vivos trust for Michael, with the...
-
Jenny purchased a home 7 years ago for $100,000, which became her primary residence. A year ago Jenny married Ethan, who moved in at that time. A few months after Ethan moved in, Jenny died in a...
-
Larisa owns a $300,000 whole life insurance G2 policy with a CSV of $42,000 and an ACB of $20,000. She applies to reduce her coverage to $200,000. Assuming her marginal tax rate is 35%, How much will...

Study smarter with the SolutionInn App


