Balance each redox reaction occurring in acidic aqueous solution. a. Zn(s) + Sn+ (aq) b. Mg(s) +
Question:
Balance each redox reaction occurring in acidic aqueous solution.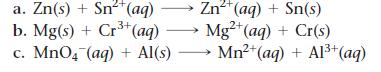
Transcribed Image Text:
a. Zn(s) + Sn²+ (aq) b. Mg(s) + Cr³+ (aq) — Zn²+ (aq) + Sn(s) Mg²+ (aq) + Cr(s) c. MnO4 (aq) + Al(s) →→→ Mn²+ (aq) + A1³+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To balance redox reactions occurring in acidic aqueous solution you can follow these steps Identify the oxidation and reduction halfreactions Balance ...View the full answer

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
PART A: Balance each redox reaction occurring in acidic aqueous solution. MnO4( a q )+Al( s )Mn2+( a q )+Al3+( a q ). Express your answer as a chemical equation. Identify all of the phases in your...
-
Balance each redox reaction occurring in acidic aqueous solution. a. I (aq) + NO (aq) b. CIO4 (aq) + Cl(aq) c. NO3(aq) + Sn+ (aq) 1(s) + NO(g) CIO3(aq) + Cl(g) Sn+ (aq) + NO(g)
-
Balance each redox reaction occurring in acidic aqueous solution. a. PbO(s) + I (aq) Pb+ (aq) + 1(s) 2+ b. SO32 (aq) + MnO4 (aq) SO42 (aq) + Mn+ (aq) 2- c. S03 (aq) + Cl(g) SO4 (aq) + Cl (aq)
-
Assume that n is a positive integer. For each of the following algorithm segments, how many times will the innermost loop be iterated when the algorithm segment is implemented and run? 1) for k:=1 to...
-
Using the data in E 10 and the average costing method, prepare a process cost report that computes the equivalent units for November, the product unit cost for the toys, and the ending balance in the...
-
Sobrero Corporation, a Mexican affiliate of a major U.S.-based hotel chain, starts the calendar year with 1 billion pesos (P) cash equity investment. It immediately acquires a refurbished hotel in...
-
Africa offers a fast growing and exciting mobile phone market, with more than 50% growth per year since 2002. More than 28% of African consumers own mobile phones; a larger market than North America....
-
Superior Markets, Inc., operates three stores in a large metropolitan area. A segmented absorption costing income statement for the company for the last quarter is given below: The North Store has...
-
Discuss each of the following terms: (a) data (b) database (c) database management system (d) database application program (e) data independence (f) security (g) integrity (h) views 2. What is...
-
Balance each redox reaction occurring in basic aqueous solution. a. HO(aq) + ClO(aq) CIO (aq) + O(g) b. Al(s) + MnO4 (aq) MnO(s) + Al(OH)4 (aq) c. Cl(g) Cl(aq) + CIO (aq)
-
How can the corrosion of iron be prevented?
-
Optically active molecules have the property of rotating the direction of polarization of linearly polarized light. Many biologically important molecules have this property, some causing a...
-
What is Registered Retirement Savings Plan (RRSP)? explain in detail
-
Kristin lives in Toronto and owns two automobiles which she insures separately. Automobile one is insured under policy A and has a liability limit of $400,000. Automobile two is insured under policy...
-
You are asked to evaluate the firm's productivity by comparing 2 year's performance. The following data is available: output (units) labor (hour) capital $ machinery $ cost of labor $ 2005 21,000...
-
5-48 A 25.400 mm, constant diameter shaft, is loaded with forces at A and B as shown, with ground reaction forces at O and C. The shaft also transmits a torque of 169.500 Nm throughout the length of...
-
A company provided the following benefits to one of its managers. Assume the exchange rate is $1=P50 and the foreign trip was for four days. How much is the fringe benefit tax for the quarter?...
-
Create a chart in which you describe the stages of Piagets theory of cognitive development. For each stage, identify a task that a child could perform.
-
The Zwatch Company manufactures trendy, high-quality moderately priced watches. As Zwatch's senior financial analyst, you are asked to recommend a method of inventory costing. The CFO will use your...
-
Express a pressure of 5.0 10 6 Pa in units of pounds per square inch.
-
An altimeter is a device used to measure altitude. Most altimeters are based on measurements of air pressure. Explain how they work and estimate how much the pressure changes for a 100-m change in...
-
The water in the pipes of a typical house is at a gauge pressure of 3.0 times atmospheric pressure. What is the absolute pressure in pascals and in units of pounds per square inch?
-
A firm books a revenue of $900,000 in December 2016. Based on previous experience, 5% of the sales are returned within 30 days. Returned products are resalable. How much should the sales return...
-
Gabriel sold CosmoVerse stock that he purchased 300 shares at $22.09 per share one year ago for a 20% loss. (Round your answer to 2 decimal places.) How much did Gabriel pay in total for their shares...
-
Brutus and Olive were married when Brutus unexpectedly died on August 4, 2015. They had both owned and lived in the same house for 25 years, which they bought for $50,000. Olive sold the house on...

Study smarter with the SolutionInn App


