Calculate the entropy of each state and rank the states in order of increasing entropy. (a) (b)
Question:
Calculate the entropy of each state and rank the states in order of increasing entropy.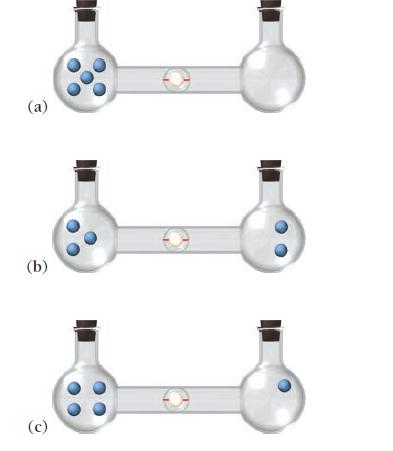
Transcribed Image Text:
(a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To calculate the entropy of each state we can use the following equation S k lnW where S is the entr...View the full answer

Answered By

Michael Mulupi
I am honest,hardworking, and determined writer
4.70+
72+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the entropy of activation for a collision between two structure less particles at 500 K, taking M = 78 g mol-3 and 0'= 0.62 nm2,
-
Rank each group of radicals in order of increasing stability: (a) (b)
-
Rank each group of compounds in order of increasing heat of hydrogenation. (a) hexa-1, 2-diene; hexa-1, 3, 5-triene; hexa-1, 3-diene; hexa-1, 4-diene; hexa-1, 5-diene; hexa-2, 4-diene. (b)
-
Where is the line in the sand the point where such behaviors are so destructive that you feel that the relationship needs to end?
-
Eagle Manufacturing supplies engine parts to Cherokee Cycle Company, a major U.S manufacturer of motorcycles. Like all of Cherokees suppliers, Eagle has always added a healthy profit margin to its...
-
How can personal selling be used to build long-term customer relationships?
-
Refer to the information in Problem 21-1B. Tohono Companys actual income statement for 2017 follows. Required 1. Prepare a flexible budget performance report for 2017. Analysis Component 2. Analyze...
-
A. Richard McCarthy (born 2/14/64; Social Security number 100-10-9090) and Christine McCarthy (born 6/1/1966; Social security number 101-21-3434) have a 19-year-old son (born 10/2/99 Social Security...
-
Student Name: Anthony Jedruczek (Please PRINT your name) Assume that Q-Caf has the following transactions related to the sale of coffee beans during the month of October, 2023. Oct 1 Oct 5 Oct 15 Oct...
-
Given the data, calculate S vap for each of the first four liquids. (S vap = H vap /T, where T is in K) All four values should be close to each other. Predict whether the last two liquids in the...
-
The solubility of AgCl(s) in water at 25 C is 1.33 * 10 -5 mol/L and its H of solution is 65.7 kJ/mol. What is its solubility at 50.0 C
-
Which phrase refers to the key choices that a company makes to bring a product service to market? a. Marketing channels b. Marketing mix c. Product life cycle d. Root cause
-
Given the following optimal LP tableau of a cost minimization problem, determine the value of the decision variables of an MIP with the following constraints: x1, x3 = +Integers x2 > 0 -Z 0 0.692308...
-
Tunneling L2 traffic over a L3 network is an example of a(n) 3-tier campus redundant star spine-leaf O overlay/underlay topology.
-
Below is the balance sheet for Loser, Inc. Loser, Inc. is an insolvent company in bankruptcy that is about to be liquidated under Chapter 7 of the bankruptcy code. All of the asset values on the...
-
Mr. Jordon Lake is an administrator for a publicly traded Canadian manufacturing company. His gross salary for the year ending December 31, 2017 is $83,000. For the 2017 taxation year, Mr. Lake's...
-
There are four main components associated with the cost of turnover: separation costs, vacancy costs, replacement costs, and training costs. Kindly explain what is the most expensive and why.
-
Complete the following, placing it in a single Word document: As a new manager in your department, you are tasked with the job of providing employee training for members of your department who have...
-
How do network effects help Facebook fend off smaller social-networking rivals? Could an online retailer doing half as much business compete on an equal footing with Amazon in terms of costs? Explain.
-
A mass m = 2.4 kg is attached to two springs as it slides along a friction less floor, while the springs are fastened to two walls as shown in Figure P11.17. The springs both have k = 400 N/m and are...
-
Estimate the spring constant for a trampoline. Assume a person is standing on the trampoline and oscillating up and down without leaving the trampoline. Begin by estimating the mass of the oscillator...
-
A mass-on-a-spring system has m = 50 kg and k = 200 N/m. The mass is pulled a distance 0.25 m from its equilibrium position and then released. (a) What is the maximum acceleration of the mass? (b)...
-
Suppose a sequence satisfies the given recurrence relation and initial conditions. Find an explicit formula for the sequence. 1. ak = 7ak-1-10ak-2, for every integer k 2 ao = -2, a = 2 2....
-
Cash and Noncash Share Issuances Skelton Corporation was organized on June 1. The company's charter authorizes 500,000 shares of $5 par value common stock. On July 1, the attorney who helped organize...
-
Consider the choice structure (B,C (.)) such that (a) B=({x,y}, {x, y, z}) and C ({x,y}) = {x}. If (B,C (.)) satisfies a weak axiom of revealed preferences (WARP), show what C ({x, y, z}) must be....

Study smarter with the SolutionInn App


