Calculate the number of copper atoms in 2.45 mol of copper. SORT You are given the amount
Question:
Calculate the number of copper atoms in 2.45 mol of copper.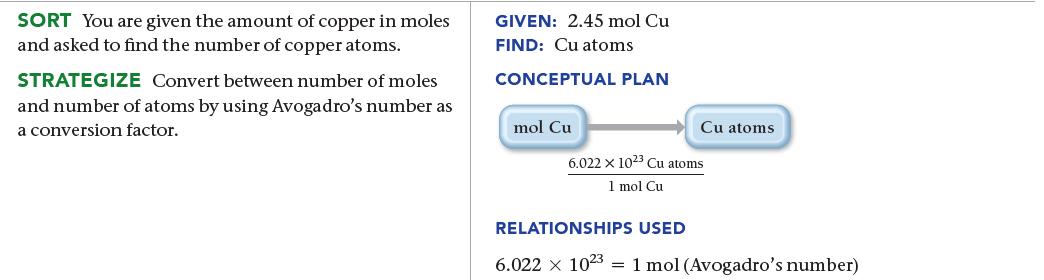
Transcribed Image Text:
SORT You are given the amount of copper in moles and asked to find the number of copper atoms. STRATEGIZE Convert between number of moles and number of atoms by using Avogadro's number as a conversion factor. GIVEN: 2.45 mol Cu FIND: Cu atoms CONCEPTUAL PLAN mol Cu Cu atoms 6.022 x 1023 Cu atoms 1 mol Cu RELATIONSHIPS USED 6.022 x 1023 1 mol (Avogadro's number)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
245 mol Cu x ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many copper atoms are in a copper penny with a mass of 3.10 g? (Assume that the penny is composed of pure copper.) SORT You are given the mass of copper and asked to find the number of copper...
-
In the experiment: Determining Avogadro's Number the values that I've got are Trial 1 Trial 2 Trial 2 Initial mass of Copper electrode (g) 12.517 12.480 12.434 Final mass of Copper electrode (g)...
-
(a) Suppose you have a cube of copper metal that is 0.236 cm on a side with a mass of 0.1206 g. If you know that each copper atom (radius = 128 pm) has a mass of 1.055 10 22 g (you will learn in...
-
At the beginning of the current season, the ledger of Highland Tennis Shop showed Cash $2,500; Inventory $1,700; and Common Stock $4,200. The following transactions were completed during April. Apr....
-
Are sunk costs ever differential costs?
-
Suppose an electron was bound to a proton, as in the hydrogen atom, but by the gravitational force rather than by the electric force, what would be the radius and energy of the first Bohr orbit?
-
In the benzene adsorber of Example 9.7, the flow rate is increased to \(0.25 \mathrm{~m}^{3} / \mathrm{s}\). Calculate the breakthrough time and the fraction of the bed adsorption capacity that has...
-
Grider Companys chart of accounts includes the following selected accounts. 101 Cash..........401 Sales 112 Accounts Receivable....414 Sales Discounts 120 Merchandise Inventory....505 Cost of Goods...
-
A jewelry shop specializes in creating gold and silver charms. The shop has five metalsmiths who work on the charms. One week, the shop's production possibilities curve shifts inward. What could...
-
As the bookkeeper for Yuvraj Cleaning Service, from the trial balance section of the worksheet on page 230, enter the beginning balance of each account before adjustments in your working papers,...
-
What are the main ideas in Daltons atomic theory? How do they help explain the laws of conservation of mass, of constant composition, and of definite proportions?
-
An argon isotope has a mass number of 40 (A = 40). How many neutrons does it contain? (a) 40 (b) 18 (c) 22
-
The following account balances are taken from the records of the Faraway Travel Agency: Faraway extends credit terms requiring full payment in 60 days, with no discount for early payment. Required 1....
-
Provide an OS Mind Map of the things you learned? Remember there is a minimum Three hundred words should be. Required Textbook: Title: Guide to Parallel Operating systems with Windows 10 and Linux...
-
Consider the following functions. f(-3)=10 and g(-3)=1 tep 1. Find (f+g)(-3)
-
In an exhaust fan, a set of gears is used to reduce the speed from a motor to the fan system (see Figure 1.1). The 20-tooth pinion gear on the motor has a diametral pitch of 5 and rotates at 1150 rpm...
-
Zack and Leah live in a community property state. Zack owns a policy on his life, the proceeds of which were paid to his daughter from a prior marriage. Zack and Leah used community funds to purchase...
-
Marc, age 67, is a single taxpayer. In 2023, he received $200,000 of interest income from an investment in city of Houston bonds and $20,000 of social security benefits. He also incurred $14,500 of...
-
Moore Entertainment sells souvenir T-shirts at each rock concert that it sponsors. The shirts cost $9 each. Any excess shirts can be returned to the manufacturer for a full refund of the purchase...
-
To help you become familiar with the accounting standards, this case is designed to take you to the FASBs Web site and have you access various publications. Access the FASBs Web site at...
-
Three of the compounds from Problem 13.14 can be prepared from the reaction between a hydride reducing agent (NaBH 4 or LAH) and a ketone or aldehyde. Identify those three compounds, and explain why...
-
For each of the following alkenes, assign the configuration of the double bond as either E or Z: a. b. c. d. F.
-
Draw the mechanism and predict the product of the following reaction. In this case, H 3 O + must be used as a proton source instead of water. Explain why. 1) xs MeMgBr 2) H*
-
HR professionals become strategic partners when they participate in the process of defi ning the business strategy, when they ask questions that move the strategy to action and when they design HR...
-
1. Prepare a presentation for your fellow students on What makes a good HR strategy? Illustrate with examples. 2. You have received an e-mail from your boss, the HR director, with the message: We...
-
Consider the sets X={0} {1/n: nN} Y = {1, 1} U {1-1/n: nN} U{-1+1/n: nN}. with the standard Euclidean metric on R. Show that X and Y are not homeomorphic, i.e. there does not exist a bijection f : XY...

Study smarter with the SolutionInn App


