Consider the reaction: The following data show the equilibrium constant for this reaction measured at several different
Question:
Consider the reaction:
![]()
The following data show the equilibrium constant for this reaction measured at several different temperatures. Use the data to find ΔH°rxn and ΔS°rxn for the reaction.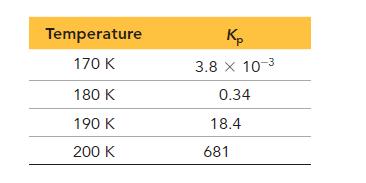
Transcribed Image Text:
2 NO(g) + O₂(g) = 2 NO₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The image you sent shows the following reaction 2 NOg O2g 2 NO2g This is the reaction of nitrogen monoxide and oxygen to form nitrogen dioxide It is a...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the reaction: The following data show the equilibrium constant for this reaction measured at several different temperatures. Use the data to find H rxn and S rxn for the reaction. H(g) +...
-
Using the FCFS rule for scheduling, the sequence is 1-2-3 For the schedule developed using the FCFS nuis, the total length of time taken to complete the three sults (including delivery) 22 hours...
-
The tabulated data show the rate constant of a reaction measured at several different temperatures. Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction....
-
An undamped mass spring system is released from rest from an initial displacement of x = 0.24 m and starts to oscillate. You see that the mass reaches its largest positive displacement for the first...
-
On June 30, New Haven Companys work in Process inventory account showed a beginning balance of $29,400. The Materials Inventory account showed a beginning balance of $240,000. Production activity for...
-
List and describe the four primary types of UPS systems. Which is the most effective and the most expensive, and why?
-
General Medical Center bought equipment on January 2, 2010, for $18,000. The equipment was expected to remain in service for four years and to perform 400 oper ations. At the end of the equipments...
-
Refer to the financial statements of American Eagle (Appendix B) and Urban Outfitters (Appendix C). Required: 1. A few years ago, American Eagle Outfitters split its stock. Describe the impact that...
-
Sandhill Company has a machine with a cost of $714000 which also is its fair value on the date the machine is leased to Pronghorn Company. The lease is for 6 years and the machine is estimated to...
-
The change in enthalpy (H rxn ) for a reaction is -25.8 kJ/mol. The equilibrium constant for the reaction is 1.4 * 10 3 at 298 K. What is the equilibrium constant for the reaction at 655 K?
-
Consider the reaction: Calculate G rxn for the reaction at 25 C under each of the following conditions: a. Standard conditions b. At equilibrium c. P CH3OH = 1.0 atm; P CO = P H2 = 0.010 atm CO(g) +...
-
What is the general path diagram for multiple regression?
-
Apple juice is being pumped at 30 C from an open tank through a sanitary pipe with an inside diameter of 25 mm to a second tank at a higher level. The mass flow rate is 1 kg/s through a 27 m of...
-
1. Draw the graph of links and change of basis diagram(s). 2. Give the vector form of the constraint equation(s) associated with the closure by a prismatic joint between 2 and 3. Develop the...
-
Multiply. 2 x +x-6 x+1 x-1 4x+12
-
Consider a combined gas - steam cycle system which has: T 3 = 5 0 0 oC , P 3 = 5 MPa, rp = 1 0 . Find net work and thermal efficiency by using Standard air analysis and Cold air analysis.
-
A bar 1 . 5 m long is made up of two parts of aluminium and steel and that cross sectional area of Aluminum bar is twice that of steel bar. The rod is subjected to an axial tensile load of 2 0 0 kN ....
-
What are the typical cash inflows from investing activities? What are the typical cash outflows from investing activities? Discuss.
-
The text defined intrinsic value as the value of an asset given a hypothetically complete understanding of the assets investment characteristics. Discuss why hypothetically is included in the...
-
Consider a Cavendish apparatus that employs a torsion fiber with k = 1.0 10 -8 N m. Suppose the smallest twist angle that can be measured is = 2.0. If the distance from one of the masses m 1 to...
-
Figure P11.62 shows the amplitude as a function of frequency for a driven, damped oscillator. Estimate the resonant frequency. Figure P11.62 ? 500 1000 1500 Frequency (Hz) Amplitude (mm)
-
Figure P11.61 shows the displacement as a function of time for an under-damped harmonic oscillator. Estimate the fraction of the mechanical energy that is ?lost? to friction during one cycle. Figure...
-
Golden State Elixirs' tinctures are produced in four steps: extraction, blending, testing, and bottling. The first step is the extraction of cannabis oil from cannabis buds, also called cannabis...
-
1. The product I have chosen is the Dell Laptop. Dell is a company that was founded in Texas and is currently head-quartered in Round Rock, Texas. They specialize in the technology industry and are...
-
The aviation industry is highly competitive and, to get noticed, you must differentiate yourself. For this reason, the Aviation School designed a flight attendant course, in which knowledge will be...
Nmap 6 Network Exploration And Security Auditing Cookbook 1st Edition - ISBN: 1849517487 - Free Book

Study smarter with the SolutionInn App


