Consider the reaction: The following data show the equilibrium constant for this reaction measured at several different
Question:
Consider the reaction:![]() The following data show the equilibrium constant for this reaction measured at several different temperatures. Use the data to find ΔH°rxn and ΔS°rxn for the reaction.
The following data show the equilibrium constant for this reaction measured at several different temperatures. Use the data to find ΔH°rxn and ΔS°rxn for the reaction.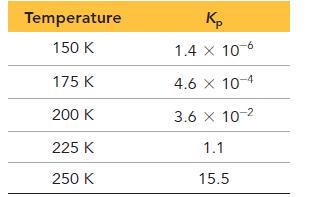
Transcribed Image Text:
H₂(g) + I₂(g) = 2 HI(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
H 50...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the reaction: The following data show the equilibrium constant for this reaction measured at several different temperatures. Use the data to find H rxn and S rxn for the reaction. 2 NO(g) +...
-
Using the FCFS rule for scheduling, the sequence is 1-2-3 For the schedule developed using the FCFS nuis, the total length of time taken to complete the three sults (including delivery) 22 hours...
-
The tabulated data show the rate constant of a reaction measured at several different temperatures. Use an Arrhenius plot to determine the activation barrier and frequency factor for the reaction....
-
8. Explain how electric forces are like gravitation forces and how they differ.
-
The three product cost elements-direct materials, direct labor, and overhead-flow through a job order costing system in a structured, orderly fashion. Specific accounts and subsidiary ledgers are...
-
Why are account balances summarized into financial statements? Why dont companies simply distribute a list of year-end account balances?
-
Thermal regeneration of a fixed-bed adsorber is based on the fact that the adsorption process is exothermic; therefore, heating the saturated adsorbent will result in desorption. The dynamics of the...
-
Zeigler Manufacturing Company purchased a robot for $720,000 at the beginning of year 1. The robot has an estimated useful life of four years and an estimated residual value of $60,000. The robot,...
-
The database contains three tables containing information about this company's sales process: Inventory, Sales, and SalesItems. Use the Relationships window to link the tables together. The...
-
The change in enthalpy (H rxn ) for a reaction is -25.8 kJ/mol. The equilibrium constant for the reaction is 1.4 * 10 3 at 298 K. What is the equilibrium constant for the reaction at 655 K?
-
Consider the reaction: Calculate G rxn for the reaction at 25 C under each of the following conditions: a. Standard conditions b. At equilibrium c. P CH3OH = 1.0 atm; P CO = P H2 = 0.010 atm CO(g) +...
-
Describe each of the following: a. Sight draft. b. Time draft. c. Bankers acceptance. d. Promissory note. e. Trade acceptance.
-
A US firm makes purchases from different parts of the world and sells them through its subsidiaries. The following is the inter-subsidiary payments matrix (i.e. Canadian subsidiary owes $36 to the US...
-
A simple approach for finding ultimate BOD (L) involved daily measurements of BoDe A straight line is fitted to a plot of BOD VS BOD on a graph paper sheet and the intersection point of that line...
-
You invest in a three-year bond with a coupon of 7% at a price of 98.5%. What is your income yield? (2) - You hold a corporate zero-coupon bond which matures (at 100%) in two years. The company pays...
-
WACC and Optimal Capital Structure F. Pierce Products Inc. is considering changing its capital structure. F. Pierce currently has no debt and no preferred stock, but it would like to add some debt to...
-
Senior Bank and Junior Bank both have security interests in the equipment of Faisal Electric. Senior Bank filed its financing statement first. On default by Faisal, Senior Bank hauls off the...
-
Exercises 9-12 refer to the following table which lists carbon dioxide concentrations (in parts per million) for each year from 1880 to 2009 with projection values for the last four years. Construct...
-
The text defined intrinsic value as the value of an asset given a hypothetically complete understanding of the assets investment characteristics. Discuss why hypothetically is included in the...
-
A damped harmonic oscillator is displaced from equilibrium and then released. The oscillator displacement as a function of time is shown in Figure P11.60. Is this oscillator under-damped or over...
-
A steel string (diameter 1.0 mm) of length 2.5 m hangs vertically. At the bottom of the string is a seat. When a child of mass m sits on the seat, she finds that the string stretches so that its new...
-
You want to use a steel cable to tow your car (m = 2000 kg). The cable is solid steel with a diameter of 1.0 cm and a length of 10 m. If you are pulling the car such that its acceleration is 1.5 m/s...
-
Your organisation has purchased a new photocopier at a cost of $17,000. The purchase was made in July so there is no need to calculate depreciation for part of the first year. The copier will be...
-
Calculate the operating income for UC Hospital using the formula and the numbers provided. Operating Revenue = $46,000,000 Operating Expenses = $38,000,000 Return on Investments = $780,000 Operating...
-
Three MBA students purchased a different asset yesterday at the price listed below. Each sold their asset today at the listed selling price. Calculate the percent return for each student rounded to...

Study smarter with the SolutionInn App


