Have each group member look up the K sp for a different compound. Calculate the molar solubility.
Question:
Have each group member look up the Ksp for a different compound.
Calculate the molar solubility. Do the numerical values suggest that the compound is soluble or insoluble?
Compare answers with the solubility rules from Chapter 5, and have each group member present his or her findings to the group.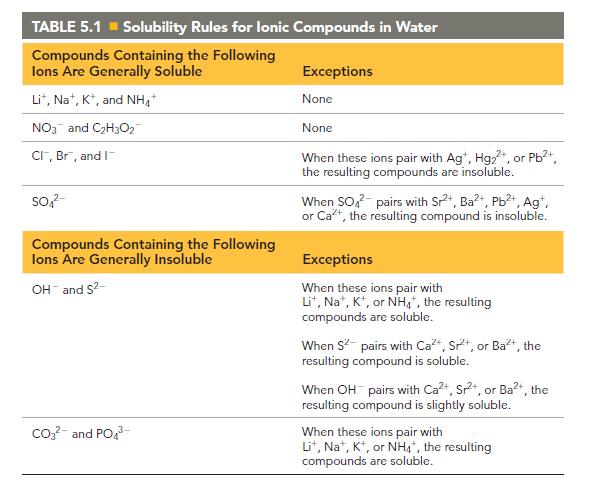
Transcribed Image Text:
TABLE 5.1 Solubility Rules for lonic Compounds in Water Compounds Containing the Following lons Are Generally Soluble Lit, Na+, K+, and NH NO3 and C₂H3O2 CI, Br, and I SO4²- Compounds Containing the Following lons Are Generally Insoluble OH and S²- CO3²- and PO4³- Exceptions None None When these ions pair with Agt, Hg₂+, or Pb²+, the resulting compounds are insoluble. When SO² pairs with Sr²+, Ba²+, Pb²+, Ag+, or Ca²+, the resulting compound is insoluble. Exceptions When these ions pair with Lit, Na+, K+, or NH4+, the resulting compounds are soluble. When S² pairs with Ca+, Sr, or Ba²+, the resulting compound is soluble. When OH pairs with Ca²+, Sr²+, or Ba²+, the resulting compound is slightly soluble. When these ions pair with Lit, Na, K, or NH4*, the resulting compounds are soluble.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Sure lets go through an example Lets say each member of the group is looking up the Ksp solubility product constant for a different compound Ill use a ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Review the solubility rules. Without referring back to the rules, have each group member list two ionic compounds that are expected to be soluble and two that are expected to be insoluble. Include at...
-
Choose a row of the transition metals in the periodic table. Have each group member look up and graph (where appropriate) a separate trend for the elements in that row, choosing from electron...
-
Assign each element in the first row of the transition metals, scandium through zinc, to a group member. Have each group member look up on the Internet or in a reputable chemistry reference book the...
-
MVP Company issued a callable bond. The bond is a 7% semiannual coupon bond currently priced at 102 that has a remaining time to maturity of seven years. The bond is callable beginning the end of...
-
Smith Accounting Services is considering a special order that it received from one of its corporate clients. The special order calls for Smith to prepare the individual tax returns of the...
-
You were recently hired as the assistant treasurer for Victor, Inc. Yesterday, the treasurer was injured in a bicycle accident and is now hospitalized, unconscious. Your boss, Mr. Fernandes, just...
-
Petitioner Christy Brzonkala met respondents Antonio Morrison and James Crawford at a campus party at Virginia Polytechnic Institute (Virginia Tech), where they were all students. At the party, the...
-
A summary of the balance sheet of Travelers Inn Inc. (TII), a company which was formed by merging a number of regional motel chains and which hopes to rival Holiday Inn on the national scene, is...
-
When controlling for many factors that determine earnings differentials (age, occupation, education, etc.), there is still an 'unexplained' difference of 11% in earnings between Black and white...
-
A base is known to be one of the three listed in the table. You are given a sample of the base and asked to identify it. To do so, you dissolve 0.30 g of the base in enough water to make 25.0 mL of...
-
With group members acting as atoms or ions, act out the reaction that occurs when HCl is added to a buffer solution composed of HC 2 H 3 O 2 and NaC 2 H 3 O 2 . Write out a script for a narrator that...
-
Recall a group of which you were a member. List each member, including yourself, and identify each persons level of status within the group. Explain why each member had the level of status you...
-
1. Show the output of the following program: #include using namespace std; int mystery (int); int main() { int n; for (n = 1; n
-
explain how organizations can apply the kotters model as a theory to managing social change
-
Nicholas owns stock in Belvidere Corporation he purchased three years ago. Nicholas sells the shares to his sister, Naomi, in the current year resulting in a realized loss to Nicholas. Number of...
-
You are given the task of reading n numbers and then printing them out in sorted order. Suppose you have access to a balanced dictionary data structure, which supports the operations search, insert,...
-
Consider the following game Player 1 T M B E 4,4 2,0 3,0 Player 2 N 0, 2 2, 2 1,0 a) Find the the pure-strategy Nash equilibria b) Find a mixed-strategy Nash equilibria in which Player 1 plays all...
-
How is Economic Darwinism a major force on industries around the globe?
-
Use the information given about the angles and to find the exact value of: (a) sin( + ) (b) cos( + ) (c) sin( - ) (d) tan ( + ) (e) sin(2) (f) cos (2) (g) sin /2 (h) cos/2 cos = 4/5, 0 < < /2; cos =...
-
A sound wave in an unknown gas has a frequency of 440 Hz and a wavelength of 2.2 m. What type of gas might it be? Use Table 13.1. TABLE 13.1 Speed of Sound in Some Common Materials Speed of Sound...
-
A battleship is using sonar (reflected underwater sound signals) to detect the presence of nearby submarines. It is found that a sonar reflection has a round-trip travel time (from the battleship to...
-
In this chapter, we discussed the Doppler effect that occurs when a source of sound moves directly toward or away from a listener. Do you think there will be a Doppler shift if the velocity of the...
-
During the 2023 FBT year, Mt Barker Brewing (MBB) provided the following benefits to employees: Laptop computer costing $1,300 Concert tickets costing $200 Water filter system costing $1,500 All...
-
Kate has an individual RESP for her daughter, Macy which was opened 20 years ago. Macy, age 32 passed away in a car accident this year. Current RESP breakdown: Contribution: $100,000 Canada Education...
-
Hamish Carter is employed as a parking attendant at Crowne Metro Hotel. During the 2023 income year, Hamish received the following amounts: Net Wages from Crowne (PAYGW of $11,000) $59,000 Tips from...

Study smarter with the SolutionInn App


