Refer to the nomenclature flowchart (Figure 3.11) to name each compound. a. HNO 2 (aq) b. B
Question:
Refer to the nomenclature flowchart (Figure 3.11) to name each compound.
a. HNO2(aq)
b. B2Cl2
c. BaCl2
d. CrCl3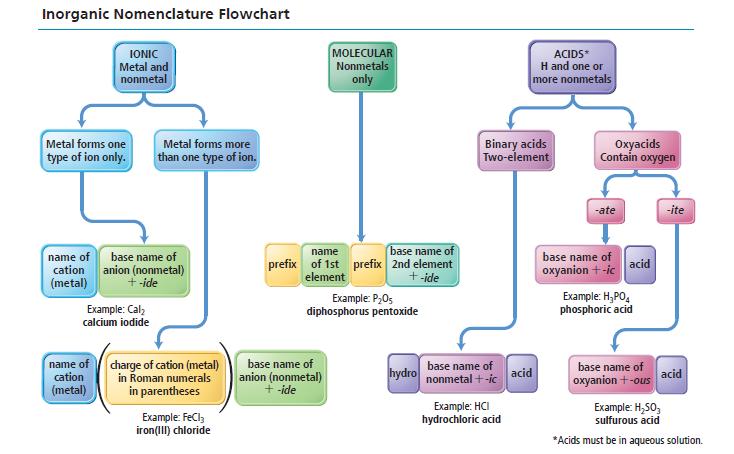
Transcribed Image Text:
Inorganic Nomenclature Flowchart IONIC Metal and nonmetal Metal forms one type of ion only. name of base name of cation anion (nonmetal) (metal) + -ide Example: Cal₂ calcium iodide name of cation (metal) Metal forms more than one type of ion. charge of cation (metal) in Roman numerals in parentheses Example: FeCl₂ iron(III) chloride MOLECULAR Nonmetals only name base name of prefix of 1st prefix 2nd element element + -ide Example: P₂05 diphosphorus pentoxide base name of anion (nonmetal) + -ide hydro Binary acids Two-element base name of nonmetal +-ic ACIDS* H and one or more nonmetals Example: HCI hydrochloric acid acid Oxyacids Contain oxygen -ate base name of oxyanion +-ic acid Example: H₂PO4 phosphoric acid base name of oxyanion + -ous -ite acid Example: H₂50₂ sulfurous acid *Acids must be in aqueous solution.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Certainly Here are the names for each compound as given using the nomenclature flo...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Refer to the nomenclature flowchart (Figure 3.11) to name each compound. a. XeO 3 b. KClO c. CoSO 4 Inorganic Nomenclature Flowchart IONIC Metal and nonmetal Metal forms one type of ion only. name...
-
Refer to the nomenclature flowchart (Figure 3.11) to name each compound. a. KClO 3 b. I 2 O 5 c. PbSO 4 Inorganic Nomenclature Flowchart IONIC Metal and nonmetal Metal forms one type of ion only....
-
Refer to the nomenclature flowchart (Figure 3.11) to name each compound. a. SrCl 2 b. SnO 2 c. P2S 5 d. HC 2 H 3 O 2 (aq) Inorganic Nomenclature Flowchart IONIC Metal and nonmetal Metal forms one...
-
In its income statement for the year ended December 31, 2020, Bramble Company reported the following condensed data. Operating expenses $754,570 Interest revenue $28,440 Cost of goods sold 1,339,800...
-
Gnomes R Us is considering a new project. The company has a debt-equity ratio of .40. The companys cost of equity is 13.50 percent, and the aftertax cost of debt is 6.25 percent. The firm feels that...
-
Murdock Paints is in the process of evaluating two mutually exclusive additions to its processing capacity. The firms financial analysts have developed pessimistic, most likely, and optimistic...
-
All of the first months activity for Crystal Clear Cleaning is as follows. Requirements 1. Journalize transactions as required from the activity data. 2. Post journal entries to T-accounts and...
-
Iris Corporation owns 30% of Fresia Corporation's stock. On November 15, Fresia Corporation, with current E & P of $320,000, distributes land (fair market value of $100,000; basis of $160,000) to...
-
On September 14th, 2023 CBSA officers in the Quebec Region seized prohibited weapons and explosive substances which led to a Montreal man being charged. An investigation was launched by the Canada...
-
T. J. Patrick is a young, successful industrial designer in Portland, Oregon, who enjoys the excitement of commodities speculation. T. J. has been dabbling in commodities since he was a teen-ager-he...
-
Write the formula for each acid. a. Phosphoric acid b. Hydrocyanic acid c. Chlorous acid
-
Write the formula for each acid. a. Hydrofluoric acid b. Hydrobromic acid c. Sulfurous acid
-
Solve each inequality in Exercises 8691 using a graphing utility. 1 x + 1 VI 2 x + 4
-
What are examples of Induced Environment threats and their sources?
-
What are examples of Natural Systems Environment threats and their sources?
-
What are examples of Human Systems Environment threats and their sources?
-
What System Elements comprise a systems Physical Environment domain?
-
Evaluate project feasibility in terms of resources, funding, and facilities.
-
Average weekly demand for free-range dog biscuits is 80 bags per week, with a standard deviation of 16 bags. Ollah uses a continuous inventory review system to manage inventory of the biscuits. Ollah...
-
In exchange for land, the company received a 12-month note on January 1. The face amount of the note is $1,000, and the stated rate of interest is 13%, compounded annually. The 13% rate is equal to...
-
A compound with molecular formula C 9 H 10 O exhibits a strong signal at 1687 cm 1 in its IR spectrum. The 1 H and 13 C NMR spectra for this compound are shown below. Identify the structure of this...
-
A compound with molecular formula C 13 H 10 O produces a strong signal at 1660 cm 1 in its IR spectrum. The 13 C NMR spectrum for this compound is shown below. Identify the structure of this...
-
A ketone with molecular formula C 9 H 18 O exhibits only one signal in its 1 H NMR spectrum. Provide a systematic (IUPAC) name for this compound.
-
During the year, Rosenberg Incorporated has the following inventory transactions. Date January 1 Transaction Beginning inventory March 4 Purchase June 9 Purchase November 11 Purchase Number of Unit...
-
Read the case study "A Team in Trouble" from Negotiation: Readings, Exercises, and Cases (NREC) on page 605. This short case describes a situation that can often be found with student teams. Students...
-
can please get some help please You've made it to the final discussion forum for HIS 132! This week we're looking at developments from the 1980s and beyond. Read the following sources: Statements of...

Study smarter with the SolutionInn App


