Without doing any calculations, determine the signs of S sys and S sur r for each chemical
Question:
Without doing any calculations, determine the signs of ΔSsys and ΔSsurr for each chemical reaction. In addition, predict under what temperatures (all temperatures, low temperatures, or high temperatures), if any, the reaction is spontaneous.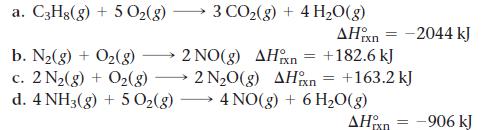
Transcribed Image Text:
a. C3H8(g) + 5 O₂(g) b. N₂(g) + O₂(g) c. 2 N₂(g) + O₂(g) d. 4 NH3(g) + 5 O₂(g) - 3 CO₂(g) + 4H₂O(g) AHixn +182.6 kJ = 2 NO(g) AH n 2 N₂O(g) AHxn = = 4 NO(g) + 6H₂O(g) -2044 kJ +163.2 kJ AH-906 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a ASsys 0 ASsurr 0 spontaneous at all tempe...View the full answer

Answered By

Salmon ouma
I am a graduate of Maseno University, I graduated with a second class honors upper division in Business administration. I have assisted many students with their academic work during my years of tutoring. That has helped me build my experience as an academic writer. I am happy to tell you that many students have benefited from my work as a writer since my work is perfect, precise, and always submitted in due time. I am able to work under very minimal or no supervision at all and be able to beat deadlines.
I have high knowledge of essay writing skills. I am also well conversant with formatting styles such as Harvard, APA, MLA, and Chicago. All that combined with my knowledge in methods of data analysis such as regression analysis, hypothesis analysis, inductive approach, and deductive approach have enabled me to assist several college and university students across the world with their academic work such as essays, thesis writing, term paper, research project, and dissertation. I have managed to help students get their work done in good time due to my dedication to writing.
5.00+
4+ Reviews
16+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Without doing any calculations, determine the signs of S sys and S sur r for each chemical reaction. In addition, predict under what temperatures (all temperatures, low temperatures, or high...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Three charges are placed as shown in the figure below. A scale is provided for the distances. Match the numerical values with the appropriate force of interaction. 19=8c a. b. 9=-2C I'm 93=4...
-
Complete the following job order cost card for six custom-built computersystems: Job Order 168 Job Order Cost Card Apache City, North Dakota Brian Patcher Batch: Custom: Computer Systems Date of...
-
What is job rotation, and what benefits does it offer an organization?
-
Lynch Brothers manufactures conveyor belts. Early in January 2011, Lynch Brothers constructed its own building at a materials, labor, and overhead cost of $900,000. Lynch Brothers also paid for...
-
Forming, the second department in a three-department production process for Chula Vista Can Inc., received 15,000 units with a total cost of $45,000 from Blanking during the month of May. Production...
-
On September 14th, 2023 CBSA officers in the Quebec Region seized prohibited weapons and explosive substances which led to a Montreal man being charged. An investigation was launched by the Canada...
-
Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. Appendix IIB a. 2 CO(g) + O(g) = 2 CO(g) b. 2 HS(g) = 2 H(g) + S(g)
-
Explain why water spontaneously freezes to form ice below 0 C even though the entropy of the water decreases during the state transition. Why is the freezing of water not spontaneous above 0 C?
-
At the end of 2013, Electric Power Associates (EPA) had total assets of $17.6 billion and total liabilities of $9 billion. Included among the assets were property, plant, and equipment with a cost of...
-
Below we present the MO diagram of ML4 molecule with a C4 point group, as well as detailed information of each MO. Here L is a o ligand that only contributes a s orbital (OL orbitals) and M is a...
-
The risk structure and the term structure of interest rate both signal following; 1. Financial Markets( Liquidity Premium Theory). 2. expectations of future economic activity.( Expectations...
-
Explain how fractional reserve banking creates money out of thin air and the potential consequences to the economy if banks over-extend themselves.. Do you think that banks should have a reserve...
-
When a market economy is operating efficiently it is also providing an equitable distribution of income and opportunity. State your opinion re: this statement. Do you agree that this is always the...
-
Assume the short run variable cost function for Japanese beer is VC=0.55 Superscript 0.8q If the fixed cost (F) is $1800 and the firm produces 550 units, determine the total cost of production (C),...
-
Harmony Company has a number of potential capital investments. Because these projects vary in nature, initial investment, and time horizon, Harmonys management is finding it difficult to compare...
-
A manufacturer can sell product 1 at a profit of $20 per unit and product 2 at a profit of $40 per unit. Three units of raw material are needed to manufacture one unit of product 1, and six units of...
-
Estimate the kinetic energy of the spinning top in Figure P9.76. Assume it is made of wood and is rotating with ? = 30 rad/s. You will need to estimate the radius of the top from the figure.? Figure...
-
Figure P12.1 shows several snapshots of a wave pulse as it travels along a string. Estimate the speed of the wave pulse. Figure P12.1 ? (m) t = 0 0.2 x (m) 10 40 50 60 70 -0.2 0.2 _t = 1 s x (m) 10...
-
Is the wave on a string in Figure P12.1 transverse or longitudinal? Figure P12.1 ? (m) t = 0 0.2 x (m) 10 40 50 60 70 -0.2 0.2 _t = 1 s x (m) 10 20 30 40 50 60 70 -0.2 _t = 2 s 0.2 x (m) 10 20 30 40...
-
Discuss the thermodynamic constraints and opportunities associated with energy storage technologies, including batteries, thermal energy storage, and hydrogen-based systems, focusing on energy...
-
The costs per equivalent unit of direct materials and conversion in the Rolling Department of Jabari Steel Company are $1.00 and $2.15, respectively. The equivalent units to be assigned costs are as...
-
Garden Sales, Incorporated, sells garden supplies. Management is planning its cash needs for the second quarter. The company usually has to borrow money during this quarter to support peak sales of...

Study smarter with the SolutionInn App


