Question: Write balanced complete ionic and net ionic equations for each reaction. a. HCl(aq) + LiOH(aq) HO(1)+ LiCI(aq) b. Cas(aq) + CuCl(aq) CuS(s) + CaCl(aq) c.
Write balanced complete ionic and net ionic equations for each reaction.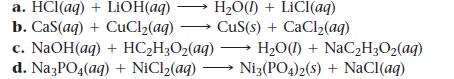
a. HCl(aq) + LiOH(aq) HO(1)+ LiCI(aq) b. Cas(aq) + CuCl(aq) CuS(s) + CaCl(aq) c. NaOH(aq) + HCHO(aq) d. Na3PO4(aq) + NiCl(aq) - HO() + NaCHO(aq) Ni3(PO4)2(s) + NaCl(aq)
Step by Step Solution
3.31 Rating (154 Votes )
There are 3 Steps involved in it
a Complete H aq Claq Lit aq OHaq Net Haq OHaq HO1 Liaq Claq HO1 b ... View full answer

Get step-by-step solutions from verified subject matter experts


