Assign oxidation numbers to all the atoms in each of the following: a. HNO3 b. CuCl c.
Question:
Assign oxidation numbers to all the atoms in each of the following:
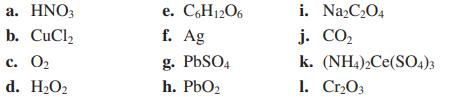
Transcribed Image Text:
a. HNO3 b. CuCl₂ c. 0₂ d. H₂O₂ e. C6H12O6 f. Ag g. PbSO4 h. PbO₂ i. Na₂C₂O4 j. CO₂ k. (NH4)2Ce(SO4)3 1. Cr₂03
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The oxidation numbers of all the atoms in each of the following compounds are a HNO3 H1 N5 O2 b CuCl ...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Assign oxidation numbers to all the atoms in each of the following: a. HNO 3 b. CuCl 2 c. O 2 d. H 2 O 2 e. C 6 H 12 O 6 f. Ag g. PbSO 4 h. PbO 2 i. Na 2 C 2 O 4 j. CO 2 k. (NH 4 ) 2 Ce(SO 4 ) 3 l....
-
Assign oxidation numbers to the atoms in each substance. a. Kr (krypton) b. krypton tetrafluoride (KrF4) c. dioxygen difluoride (O2F2)
-
Assign oxidation numbers to the atoms in each substance. a. Lithium hydride (LiH) b. Potassium peroxide (K2O2) c. Potassium fluoride (KF)
-
I recently heard my neighbor discussing how one of our other neighbors lost his job. My neighbor assumed that the person who lost his job was probably lazy or not smart enough for the job, without...
-
Prepare the journal entries to record these transactions on Kesler Companys books using a periodic inventory system. (a) On March 2, Kesler Company purchased $800,000 of merchandise from Rice...
-
A tree with n vertices has n 1 edges (Proof by induction).
-
Dr. Walter Sullivan was one of several plastic surgeons in Las Vegas visited by Julie Jones. Jones, an exotic dancer, sought plastic surgery to improve her ability to make money in her profession....
-
Sekhon Company had a beginning inventory on January 1 of 160 units of Product 4-18-15 at a cost of $20 per unit. During the year, the following purchases were made. 1,000 units were sold. Sekhon...
-
What is a sampling distribution? Explain
-
In the electrolysis of an aqueous solution of Na 2 SO 4 , what reactions occur at the anode and the cathode (assuming standard conditions)? 2- S0 +2e 250 O + 4H+ + 4e 2HO 2HO + 2e H + 2OH Na + e Na...
-
(a) How is resistance present in all transmission lines? (b) How is inductance present in all transmission lines? (c) How is capacitance present in all transmission lines? (d) The combined effects of...
-
Novak SpA is preparing its 2022 statement of cash flows, using the indirect method. The following is a list of items that may affect the statement. Using the code letters provided, indicate how each...
-
Theories on Developing Children's Cooperative Behaviour Current theory Summary Source Publication date Emerging theory Summary Source Publication date Research on Developing Children's Cooperative...
-
The founders are worried about the consequences of being in breach of contract. Advise Delightful Developers of the basic elements required for the formation of a contract and the basis on which...
-
1- Explain how productivity is measured in (the training center). 2- when you need to hire new employee How we can measure Overtime Hours for new employee? 2- Name and elaborate on two fixed costs...
-
1. Axiomatic sematics has two distinct applications: program verification and program sematics specifications. What is axiomatic sematic based on? 2. Describe the general approach to use axiomatic...
-
Reflection 2 | Algorithmic Bias Understanding and addressing algorithmic bias is crucial to prevent AI from perpetuating societal biases and discrimination through their data, algorithms, and...
-
(a) Your friend Katie Fehr cannot understand how the characteristic of corporation management is both an advantage and a disadvantage. Clarify this problem for Katie. (b) Identify and explain two...
-
Find the reduced echelon form of each of the matrices given in Problems 120. c 1 26 + 4
-
Can a molecule with an inversion center have a dipole moment? Give an example of a molecule with this symmetry element and explain your reasoning.
-
Which of the three normal modes of H 2 O in Figure 27.9 is best described as a bending mode? Does the bond angle remain unchanged in any of the modes? Which requires less energy, bond bending or bond...
-
Why does the list of elements for the D 6h group in Table 27.2 not list the elements C 2 6 , C 3 6 , and C 4 6 ? Selected Point Groups and Their Elements Symmetry Elements TABLE 27.2 Example Molecule...
-
You are a local solicitor in a suburb in which two young tax agents wish to establish a new partnership. They seek your advice on whether they or their staff could be sued by clients if the...
-
CMS pays a 10 % bonus to physicians who provide Medicare-covered services in geographic areas that lack enough health care providers to meet the health care needs of the population, Health...
-
Boulder got into a fight with Smyth and fractured Smyth's jaw. Smyth sued Boulder for his injuries. Would this case be heard in a trial or an appellate court? Why?

Study smarter with the SolutionInn App


