In the electrolysis of an aqueous solution of Na 2 SO 4 , what reactions occur at
Question:
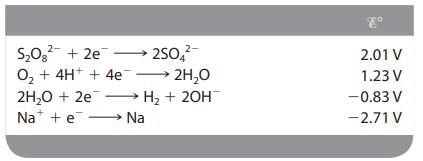
In the electrolysis of an aqueous solution of Na2SO4, what reactions occur at the anode and the cathode (assuming standard conditions)?
Transcribed Image Text:
2- S₂0² +2e 250 O₂ + 4H+ + 4e →→→ 2H₂O 2H₂O + 2e → H₂ + 2OH Na + e→→→→→→→→→ Na 2.01 V 1.23 V -0.83 V -2.71 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
In the electrolysis of an aqu...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write equations for the half-reactions that occur at the anode and cathode in the electrolysis of molten KBr. What are the products formed at the anode and cathode in the electrolysis of aqueous KBr?
-
Which product, O 2 or F 2 , is more likely to form at the anode in the electrolysis of an aqueous solution of KF? Explain your reasoning.
-
Predict the products formed in the electrolysis of an aqueous solution of CdSO 4 .
-
Using Figure 7-5 as an example, redraw Figure 7-12 using an enterprise information system that processes a shared database. Explain the advantages of this system over the paper-based system in Figure...
-
On October 5, Lane Company buys merchandise on account from OBrien Company. The selling price of the goods is $5,000, and the cost to OBrien Company is $3,000. On October 8, Lane returns defective...
-
(a) Two of the eigenvectors of a vibrating system are known to be Prove that these are orthogonal with respect to the mass matrix \[[m]=\left[\begin{array}{lll}1 & 0 & 0 \\0 & 2 & 0 \\0 & 0 &...
-
Cite several significant events in the development of contemporary internal auditing in the U.S.
-
(Single-step Income Statement) The financial records of LeRoi Jones Inc. were destroyed by fire at the end of 2004. Fortunately the controller had kept certain statistical data related to the income...
-
13. Now two forces are applied to the cart with two different spring scales as shown below. The spring scale FA still reads 10.5 N. FB The cart now moves toward the right with an acceleration toward...
-
K Dhoni is a business consultant. The following trial balance is provided at 30 September 201: The following additional information is supplied: 1. At 30 September 201 rent received in advance...
-
Gold is produced electrochemically from an aqueous solution of Au(CN) 2 - containing an excess of CN - . Gold metal and oxygen gas are produced at the electrodes. What amount (moles) of O 2 will be...
-
Assign oxidation numbers to all the atoms in each of the following: a. HNO3 b. CuCl c. 0 d. HO e. C6H12O6 f. Ag g. PbSO4 h. PbO i. NaCO4 j. CO k. (NH4)2Ce(SO4)3 1. Cr03
-
Mara, age 32, earns $60,000 working in 2022. She has no other income. Her medical expenses for the year total $6,000. During the year, she suffers a casualty loss of $9,500 when her apartment is...
-
In 2023, Kathleen Tweardy incurs $30,000 of interest expense related to her investments. Her investment income includes $7,500 of interest, $6,000 of qualified dividends, and a $12,000 net capital...
-
O&G Company manufactures console tables and uses an activity-based costing system to allocate all manufacturing conversion costs. Each console table consists of 40 separate parts totaling $210 in...
-
HomeLife Life Insurance Company has two service departments (actuarial and premium rating) and two production departments (advertising and sales). The distribution of each service department's...
-
The following account balances at the beginning of January were selected from the general ledger of Superior Juice Manufacturing Company: Work in process inventory Raw materials inventory Finished...
-
Oslo Company prepared the following contribution format income statement based on a sales volume of 1,000 units (the relevant range of production is 500 units to 1,500 units): Sales Variable expenses...
-
Apple Inc. has established a unique reputation in the consumer electronics industry with its development of products such as the iPod, the iPhone, and the iPad. As of May 2010, Apple had surpassed...
-
Separate variables and use partial fractions to solve the initial value problems in Problems 18. Use either the exact solution or a computer-generated slope field to sketch the graphs of several...
-
A surface for which the electrostatic potential is negative delineates regions in a molecule that are subject to electrophilic attack. It can help you to rationalize the widely different chemistry of...
-
Hydrocarbons are generally considered to be nonpolar or weakly polar at best, characterized by dipole moments that are typically only a few tenths of a debye. For comparison, dipole moments for...
-
Chemists know that nitric and sulfuric acids are strong acids and that acetic acid is a weak acid. They would also agree that ethanol is at best a very weak acid. Acid strength is given directly by...
-
Cantor Company had 600 units of product in its Work in Process inventory at the beginning of the period. During the period Cantor started 4,300 additional units of product. At the end of the period...
-
Mickley Company's plantwide predetermined overhead rate is $21.00 per direct labor-hour and its direct labor wage rate is $13.00 per hour. The following information pertains to Job A-500: Direct...
-
Valley's managers have made the following additional assumptions and estimates: Estimated sales for July and August are $345,000 and $315,000 respectively Each month's sales are 20% cash sales and...

Study smarter with the SolutionInn App


