Consider the following half-reactions: (a) Choosing from among the reactants in these half-reactions, identify the strongest and
Question:
Consider the following half-reactions: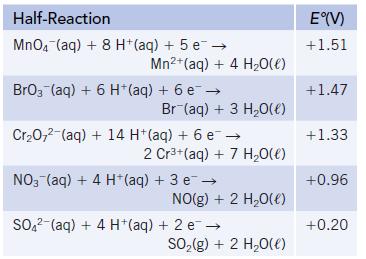
(a) Choosing from among the reactants in these half-reactions, identify the strongest and weakest oxidizing agents.
(b) Which of the oxidizing agents listed is (are) capable of oxidizing Br−(aq) to BrO3−(aq) (in acid solution)?
(c) Write a balanced chemical equation for the reaction of Cr2O72−(aq) with SO2(g) in acid solution. Is this reaction product-favored or reactant-favored at equilibrium?
(d) Write a balanced chemical equation for the reaction of Cr2O72−(aq) with Mn2+(aq). Is this reaction product-favored or reactant-favored at equilibrium?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





