Make the following temperature conversions: C (a) (b) 63 (c) K 77 1450
Question:
Make the following temperature conversions: 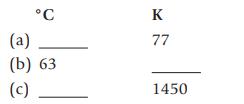
Transcribed Image Text:
°C (a) (b) 63 (c) K 77 1450
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
a Convert 77F to C To convert from Fahrenheit to Celsius you can use the formula C F 3...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Sulfuric acid is the chemical produced in the United States with the highest volume of production. In one of the earliest processes used to make it, an ore containing iron pyrites (FeS 2 ) is roasted...
-
Write a program that uses a two-dimensional array to store the highest and lowest temperatures for each month of the year. The program should output the average high, average low, and the highest and...
-
The following table presents the mid-rates, the Royal Banks buy and sell rates, and ICEs buy and sell rates (at their Vancouver International Airport operation) on the same date for conversions...
-
Molybdenum forms a substitutional solid solution with tungsten. Compute the weight percent of molybdenum that must be added to tungsten to yield an alloy that contains 1.0 10 22 Mo atoms per cubic...
-
List any errors you can find in the following partial balancesheet. Pembroke Company Balance Sheet July 31, 2006 Assets Current assets: Cash $ 43,750 18000 282,000 71,200 647,380 eivable Plus...
-
On July 1, Year 1, Danzer Industries Inc. issued $40,000,000 of 10-year, 7% bonds at a market (effective) interest rate of 8%, receiving cash of $37,282,062. Interest on the bonds is payable...
-
The personal training business owned by David Hella made the following cash transactions during the reporting period. Ignore GST. Required (a) Prepare a statement of cash flows using the direct...
-
Harte Systems, Inc., a maker of electronic surveillance equipment, is considering selling to a well-known hardware chain the rights to market its home security system. The proposed deal calls for the...
-
The functions fand g are defined as follows. f(x)=x-6x-4 and g(x)=- x-4 3x-5 Find f(x+8) and g (4). Write your answers without parentheses and simplify them as much as possible. f(x+8)= *(4)-0 g = H 5
-
Many laboratories use 25 C as a standard temperature. What is this temperature in kelvins?
-
The temperature on the surface of the Sun is 5.5 10 3 C. What is this temperature in kelvins?
-
If you were to determine the P/O ratio for oxidation of a-ketoglutarate, you would probably include some malonate in your reaction system. Why? Under these conditions, what P/O ratio would you expect...
-
Corporation is a company which handles the auction of the different antiques. The company wants to shift from the offline system to the online auction system. For this they have consulted a database...
-
How does Windows 11 process and threads work in process management
-
Identify one of the five paradoxes described in our readings and provide an example of the one you selected. Therein lies the primary paradoxbalancing individual differences and goals AND the...
-
2- An elastic spring of force constant, k = 5x10 N/m is compressed by, x = 2x10-2 m. A block at the end of the compressed spring is released from rest, see the figure. (a) Find the force to compress...
-
The mass of the Moon is approximately 7.35 E+22 kg, and its mean distance form the Earth is about 3.80 E+8 km. Determine the force of mutual gravitational attraction in newtons between the Earth and...
-
Required Identify which of the following items are added to or subtracted from the unadjusted bank balance to arrive at the true cash balance. Distinguish the additions from the subtractions by...
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
Propose a plausible synthesis for the following transformation. Enantiomer
-
Using bromobenzene and ethylene oxide as your only sources of carbon, show how you could prepare trans- 1, 2-diphenyloxirane (a racemic mixture of enantiomers). + Enantiomer
-
The S N 2 reaction between a Grignard reagent and an epoxide works reasonably well when the epoxide is ethylene oxide. However, when the epoxide is substituted with groups that provide steric...
-
If Pr[A]=0.4, Pr[B] = 0.3 and Pr[A B] = 0.18. a. Are A, B independent events? Justify your answer. (Hint: find Pr [A] first) b. Find Pr [AUB] c. Find Pr [A'|B]. Hint: A' n B = B-A
-
Develop a shaping strategy. The change that may occur will entail changing some element of performancestructure, processes, technology, groups, or the task. The purpose of the strategy is to...
-
Suppose that the current market price of the bond in Problem 1 is 95. Calculate the resulting redemption yield. A corporate bond with exactly five years left to maturity pays interest at 8% on its...

Study smarter with the SolutionInn App


