The density of pure water at various temperatures is given below Suppose your laboratory partner tells you
Question:
The density of pure water at various temperatures is given below 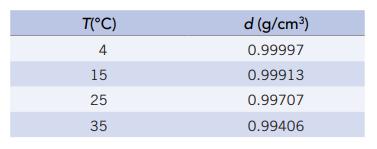
Suppose your laboratory partner tells you the density of water at 20°C is 0.99910 g/cm3. Is this a reasonable number? Why or why not?
Transcribed Image Text:
T(°C) 4 15 25 35 d (g/cm³) 0.99997 0.99913 0.99707 0.99406
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
To determine whether the reported density of water at 20C 099910 gcm is reaso...View the full answer

Answered By

Salmon ouma
I am a graduate of Maseno University, I graduated with a second class honors upper division in Business administration. I have assisted many students with their academic work during my years of tutoring. That has helped me build my experience as an academic writer. I am happy to tell you that many students have benefited from my work as a writer since my work is perfect, precise, and always submitted in due time. I am able to work under very minimal or no supervision at all and be able to beat deadlines.
I have high knowledge of essay writing skills. I am also well conversant with formatting styles such as Harvard, APA, MLA, and Chicago. All that combined with my knowledge in methods of data analysis such as regression analysis, hypothesis analysis, inductive approach, and deductive approach have enabled me to assist several college and university students across the world with their academic work such as essays, thesis writing, term paper, research project, and dissertation. I have managed to help students get their work done in good time due to my dedication to writing.
5.00+
4+ Reviews
16+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
A look at the harmful effects of too much fast food is an interesting question as to whether the fastfood restaurants should be held liable for those health problems Do you agree or disagree Why or...
-
The vapor pressure of water at various temperatures is in Table E11-11: (a) Draw a scatter diagram of these data. What type of relationship seems appropriate in relating y to x? (b) Fit a simple...
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
You have just completed your four-year degree at LLC University! Your student loans that you have accumulated while studying at LLC total $25,000. Since you have graduated, you must now begin...
-
1. What are the ethical issues involved in pizza deliveries to dangerous neighborhoods that are often predominantly inhabited by minorities? What tensions exist between economic and ethical issues?...
-
What are two processes companies can use to search for differentiation opportunities?
-
Baxter Manufacturing Ltd has determined total factory overhead costs at both minimum and maximum levels of production 100 000 machine hours and 150 000 machine hours respectively. The total factory...
-
A producer of picture frames uses a tracking signal with limits of ±4 to decide whether a forecast should be reviewed. Given historical information for the past four weeks, compute the...
-
On May 1, Soriano Company reported the following account balances along with their estimated fair values: Receivables Inventory Copyrights Account Patented technology Total assets Current liabilities...
-
When you heat popcorn, it pops because it loses water explosively. Assume a kernel of corn, with a mass of 0.125 g, has a mass of only 0.106 g after popping. (a) What percentage of its mass did the...
-
Diamond has a density of 3.513 g/cm 3 . The mass of diamonds is often measured in carats, where 1 carat equals 0.200 g. What is the volume (in cubic centimeters) of a 1.50-carat diamond?
-
Solve the linear programming model formulated in Problem 35 for Alexis Harrington graphically. a. How much would the return for cattle have to increase in order for Alexis to invest only in cattle?...
-
An initial investment of $1,000 in an account grows to $1,477.46 at the end of eight years. If the annually compounded interest rate is unchanged over the life of the investment, What is the annual...
-
Compute the Cost of Goods Manufactured and Cost of Goods Sold for South Marine Company for the most recent year using the amounts described next. Assume that Raw Materials Inventory contains only...
-
As an advocate for play in the classroom, you are going to design a training workshop that will be presented to fellow teachers about: The importance of play How play contributes to social and...
-
Danube Company purchased a used machine for $10,000. The machine required installation costs of $6,000 and insurance while in transit of $600. At which of the following amounts would the machine be...
-
1. A Bates motor is a solid rocket motor with grains cast into multiple separate segments, each a cylindrical shape as shown. (Actually the sides taper in slightly, so the cross section is really...
-
The following information pertains to Stanley Company for 2016: Beginning inventory ..... 90 units @ $15 Units purchased ......320 units @ $19 Ending inventory consisted of 40 units. Stanley sold 370...
-
Consider a game of poker being played with a standard 52-card deck (four suits, each of which has 13 different denominations of cards). At a certain point in the game, six cards have been exposed. Of...
-
In Section 4.2, we learned how to name bicyclic compounds. Using those rules, together with the rules discussed in this section, provide a systematic name for the following bicyclic compound:
-
Classify each of the following alkenes as mono-substituted, di-substituted, tri-substituted, or tetra-substituted: a. b. c. d. e.
-
Three of the compounds from Problem 13.14 can be prepared from the reaction between a hydride reducing agent (NaBH 4 or LAH) and a ketone or aldehyde. Identify those three compounds, and explain why...
-
Cost of Production Report Hana Coffee Company roasts and packs coffee beans. The process begins by placing coffee beans into the Roasting Department. From the Roasting Department, coffee beans are...
-
The BIG Idea Although credit abuse can cause serious financial problems, credit is an important part of our economy. What are advantages of allowing people and businesses to borrow money? Also...
-
The following information has been taken from the ledger accounts of Indigo Corporation. Total income since incorporation $309,000 Total cash dividends paid 55,000 Total value of stock dividends...

Study smarter with the SolutionInn App


