The equation for the reaction of aluminum and bromine is If you use 6.0 10 23
Question:
The equation for the reaction of aluminum and bromine is ![]() If you use 6.0 × 1023 molecules of Br2 in a reaction how many atoms of Al will be consumed?
If you use 6.0 × 1023 molecules of Br2 in a reaction how many atoms of Al will be consumed?
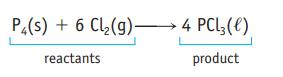
Transcribed Image Text:
2 Al(s) + 3 Br₂(l) → Al₂Bro(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
To determine how many atoms of aluminum Al will be consumed in the reaction of 2 Als 3 Br2l 2 AlBr3s ...View the full answer

Answered By

AJIN KURIAKOSE
I HAVE ELECTRONICS ENGINEERING DEGREE..AND MY AREA OF INTEREST IS MATHEMATICS,CONTROL SYSTEM,NETWORK,DIGITAL
4.70+
21+ Reviews
32+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Terence purchased a whole life policy in 1977, which he later transferred to his wife Gertrude in 1989. What group would Gertrude's policy fall under for tax purposes? a) G1 b) G2 c) G3 d) G4
-
The equilibrium-constant expression for a reaction is What is the equilibrium-constant expression when the equation for this reaction is halved and then reversed? NO2O IN20512
-
The equilibrium-constant expression for a reaction is What is the equilibrium-constant expression when the equation for this reaction is halved and then reversed? [NH,110,1 K. [NO]'[H,O1
-
The annual consumption of beef per person was about 64 7 lb in 2000 and about 60 3 lb in 2007 Assume B(t), the annual beef consumption t years after 2000, is decreasing according to the exponential...
-
A small country can import a good at a world price of 10 per unit. The domestic supply curve of the good is S = 20 + 10P The demand curve is D = 400 5P In addition, each unit of production yields a...
-
When you colored the multiples of 2, the number 4 was the first multiple of 2 that fell through the sieve. When you colored the multiples of 3, the number 9 was the first multiple of 3 that fell...
-
A hydraulic press has a ram diameter of 250 mm and a plunger diameter of 30 mm. For a force of 40 N applied on the plunger, find the force exerted on the object by the hydraulic press.
-
The following summary transactions occurred during 2013 for Bluebonnet Bakers: Cash Received from: Customers....................$380,000 Interest on note receivable .............6,000 Principal on...
-
The department head has the impression that some employees are less satisfied with their manager if the employee has worked in other companies. What is the correlation between Number of Companies...
-
Oxidation of 1.00 g of carbon monoxide, CO, produces 1.57 g of carbon dioxide, CO 2 . How many grams of oxygen were required in this reaction? P4(s) + 6 Cl(g) 4 PCL3 (1) reactants product
-
Which of the following compounds has the highest mass percent of chlorine? (a) BCl 3 (b) AsCl 3 (c) GaCl 3 (d) AlCl 3 (e) PCl 3
-
Determine the distance traveled by the spacecraft of Prob. 14.99 during the rocket engine firing, knowing that its initial speed was 7500 ft/s and the duration of the firing was 60s.
-
Here are the returns on two stocks: Digital Cheese Executive Fruit January +18 +8 February -4 +1 March +6 +5 April +8 +16 may -5 +2 June +4 +6 July -3 -4 August -9 -3 A-1) Calculate the variance...
-
The Blast company prices GC game consoles at $450 per unit. GameCore sells the GC's at $675. Annual demand at this retail price turns out to be 250,000 units. GameCore incurs ordering, receiving, and...
-
What might taking a stance against his CEO mean for Ankit's career at IvoryTower and beyond? (can from case or personal idea) 60-100 words Ankits deputy Case Study Should We Embrace Crypto? by...
-
You are approached to comment on a company's accounting disclosure quality. As an accounting analyst, what are the factors will you consider in your assessment and why?
-
Writing a proposal for cryptocurrency usage in retail industry. Can anyone please suggest me some good resources...what sort of research studies i have to perform for my dissertation. Is explorative...
-
A parent company acquired an 80% interest in a subsidiary on July 1, 2011. The subsidiary closed its books on that date. The subsidiary reported net income of $60,000 for 2011, earned evenly during...
-
In each of the following independent cases, document the system using whatever technique(s) your instructor specifies. a. Dreambox Creations (www.dreamboxcreations.com/) in Diamond Bar, California,...
-
For each of the compounds below, locate the pattern we just learned (lone pair next to a Ï bond) and draw the appropriate resonance structure: a. b. c. d. e. f. g. h. NH2
-
Draw the resonance structure(s) for each of the compounds below: a. b. c. d.
-
The formalism of the Youngs modulus is sometimes used to calculate the reversible work involved in extending or compressing an elastic material. Assume a force F is applied to an elastic rod of...
-
Calculate the time complexity for the following function in terms of Big O notation. Explain your answer. [Note: Line numbers are only for reference purpose] int fun( int n) { 1 int count = 0; 2 for...
-
Write the complete code to remove the node "banana" in the singly linked list shown in the Fig. 1. The sample code for the class Node is given below: public class Node { public String element; public...
-
An AVL tree is a binary search tree that is height balanced: for each node x, the heights of the left and right subtrees of x differ by at most 1. To implement an AVL tree, we maintain an extra...

Study smarter with the SolutionInn App


