The separation between carbon atoms in diamond is 0.154 nm. What is their separation in meters? In
Question:
The separation between carbon atoms in diamond is 0.154 nm. What is their separation in meters? In picometers (pm)? In Angstroms (Å)? 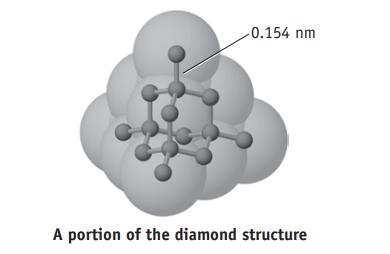
Transcribed Image Text:
-0.154 nm A portion of the diamond structure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
To find the separation between carbon atoms in diamond in ...View the full answer

Answered By

Muhammad Khurram
I have strong General Management skills to apply in your projects. Over last 3 years, I have acquired great knowledge of Accounting, Auditing, Microsoft Excel, Microsoft PowerPoint, Finance, Microsoft Project, Taxation, Strategic Management, Human Resource, Financial Planning, Business Planning, Microsoft Word, International Business, Entrepreneurship, General Management, Business Mathematics, Advertising, Marketing, Supply Chain, and E-commerce. I can guarantee professional services with accuracy.
4.80+
249+ Reviews
407+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The potential energy of two atoms in a diatomic molecule is approximated by V(r) = a/r12 - b/r2, where r is the spacing between atoms and a and b are positive constants. (a) Find the force F( r) on...
-
"Welcome to Hudson Jewelers, the Naples, Florida jewelry store attendant said with a smile. Im Bill; let me know if I can answer any questions you may have. Im browsing with my fianc, the customer...
-
An X-ray beam with = 154 pm incident on the surface of a crystal produced a maximum reflection at an angle of =28.3 . Assuming n = 1, calculate the separation between layers of atoms in the crystal.
-
Determine the degrees of freedom under the following conditions: (a) Tl-20 wt% Pb at 325 C and 400 C; (b) Tl-40 wt% Pb at 325 C and 400 C; (c) Tl-90 wt% Pb at 325 C and 400 C. Refer to the phase...
-
What can we learn about business ethics from the recent financial crisis?
-
On December 31, 2019, the balances of the accounts appearing in the ledger of Wyman Company are as follows: Instructions 1. Does Wyman Company use a periodic or perpetual inventory system? Explain....
-
Information from the records of Powertools Pty Ltd for the year ended 30 June 2025 is given below. Required Calculate the ending work in process inventory on 30 June 2025. Factory overhead, 150% of...
-
Refer to the data in Exercise. In Exercise, Back Mountain Industries (BMI) has two divisions: East and West. BMI has a cost-of-capital of 15%. Selected financial information (in thousands of dollars)...
-
Multiply and leave your answer in scientific notation. (2 105) - (4 10) =
-
A red blood cell has a diameter of 7.5 m (micrometers). What is this dimension in (a). Meters, (b). Nanometers, and (c). Picometers?
-
Carbon tetrachloride, CCl 4 , a common liquid compound, has a density of 1.58 g/cm 3 . If you place a piece of a plastic soda bottle (d = 1.37 g/cm 3 ) and a piece of aluminum (d = 2.70 g/cm 3 ) in...
-
Rod AB can slide freely along the floor and the inclined plane. At the instant shown, the velocity of end A is 1.4 m/s to the left. Determine (a) The angular velocity of the rod, (b) The velocity of...
-
You need to calculate the completed and removed from WIP Inventory - Mixing. Costs in beginning WIP inventory $1,700 Additional costs to finish units in beginning WIP inventory $400 Costs of units...
-
An electron is separated from a proton by a distance of 0.53 A. What is the valu of electric field at the location of the electron?
-
Computing the necessary sample size for an interval estimate of a population proportion requires a planning value for p. In case of any uncertainty about an appropriate planning value, we know the...
-
You are casting high carbon steel and need a 50:1 atomic ratio of Fe:C (fifty times as many Fe atoms as C atoms) in the powder you will melt and cast. If you use 2.3 kg (2300 g) of Fe powder, what is...
-
Risk factors can lead to adversity. Protective factors can lead to resilience. We've learned that ACEs are common. So is resilience! We know that the research on ACEs is FACT, but the statistics...
-
Vulcan Service Co. experienced the following transactions for 2016, its first year of operations: 1. Provided $91,000 of services on account. 2. Collected $72,000 cash from accounts receivable. 3....
-
Discuss whether responsible human resources management should apply different standards for the home company and suppliers, for developed countries and developing countries, and for large companies...
-
(R)-Limonene is found in many citrus fruits, including oranges and lemons: Draw the structures and identify the relationship of the two products obtained when (R)-limonene is treated with excess...
-
Consider the structures of cis-decalin and trans-decalin: (a) Which of these compounds would you expect to be more stable? (b) One of these two compounds is incapable of ring flipping. Identify it...
-
Atorvastatin is sold under the trade name Lipitor and is used for lowering cholesterol. Annual global sales of this compound exceed $13 billion. Assign a configuration to each chirality center in...
-
A national advertising program that costs $200,000 today is expected return a profit (like interest) of $72,000 in 6 years. Determine: (4 marks) a. The rate of return (interest) using simple...
-
xPlease address the questions below in a memorandum to Apex Investment Partners. Please include your revised capitalization tables and waterfall diagrams. How, if at all, would you change the...
-
Sky Company employed Tom Mills in Year 1. Tom earned $5,700 per month and worked the entire year. Assume the Social Security tax rate is 6 percent for the first $110,000 of earnings, and the Medicare...

Study smarter with the SolutionInn App


