Calcium carbonate (limestone, CaCO 3 ) dissolves in hydrochloric acid, producing water and carbon dioxide. An unbalanced
Question:
Calcium carbonate (limestone, CaCO3) dissolves in hydrochloric acid, producing water and carbon dioxide. An unbalanced net ionic equation for this reaction is given below. Balance it.
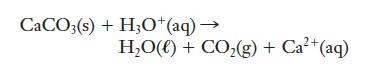
Transcribed Image Text:
CaCO3(s) + H3O+ (aq) → H,O() + CO,(g)+Ca+(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Balanced chemical equation ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Calcium carbonate (limestone, CaCO 3 ) dissolves in hydrochloric acid, producing water and carbon dioxide as shown in the following unbalanced net ionic equation: Suppose 5.0 g of CaCO 3 is added to...
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
9. If a two-dimensional array is declared as int M[3][2]; and it has been initialized to all zero's, what will be the contents of this two-dimensional array after executing the code fragment below...
-
A 10.231-g sample of window cleaner containing ammonia was diluted with 39.466 g of water. Then 4.373 g of solution were titrated with 14.22 mL of 0.1063 M HCl to reach a bromocresol green end point....
-
A firm wishing to evaluate interest rate behavior has gathered data on the nominal rate of interest and on inflationary expectations for five U.S. Treasury securities, each having a different...
-
Which is the better investment and why: 8% compounded quarterly or 8.3% compounded annually?
-
The Dewey Construction Company processes its payroll transactions to update both its payroll master file and its work-in-process master file in the same computer run. Both the payroll master file and...
-
Anne is employed by Bradley Contracting Company. Bradley has a $1.3 million contract to build a small group of outbuildings in a national park. Anne alleges that Bradley Contracting has discriminated...
-
Draw a waveform for the A/D successive approximation method as shown in Fig. 5, if the input voltage of the comparator is considered to be 5.5 V. +16 V ko 2R WK5 V 5.5 V OFF 1k0 2R 1ka 21 DoD 2R w 1...
-
Methyl cyanoacrylate is the chemical name for the substance sold as Super Glue, and it has the chemical formula C 5 H 5 NO 2 . Calculate the number of molecules of this substance in a 1.0-ounce tube...
-
Many chemical reactions take place in the catalytic converter of a car. In one of these reactions, nitric oxide (NO)reacts with ammonia (NH 3 ) to give nitrogen (N 2 ) and water. Write a balanced...
-
In Figure a uniform rod of mass m is hinged to a building at its lower end, while its upper end is held in place by a rope attached to the wall. If angle 1 = 60, what value must angle 2 have so that...
-
The vector of forces for the right-hand side of the equations defining the principal coordinates is calculated by \(\mathbf{G}=\mathbf{P}^{T} \mathbf{F}\). Indicate whether the statement presented is...
-
The elements of the impedance matrix are the transfer functions \(G_{i, j}(s)\). Indicate whether the statement presented is true or false. If true, state why. If false, rewrite the statement to make...
-
For a system with a damping matrix that is proportional to the stiffness matrix, the higher modes are more highly damped and therefore have less of an effect on the forced response. Indicate whether...
-
The Laplace transform method cannot be used to determine the response of a system with proportional damping. Indicate whether the statement presented is true or false. If true, state why. If false,...
-
Why is the impulsive response of a system with motion input not defined?
-
Example 1 used measured performance IQ scores for three different blood lead levels. If we use the same three categories of blood lead levels with the full IQ scores, we get the accompanying Excel...
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
Draw a mechanism for each of the following transformations: a. b. c. ,t Dilute H,SO,
-
In this problem, you calculate the error in assuming that ÎH o R is independent of T for the reaction 2CuO(s) 2Cu(s) + O 2 (g). The following data are given at 25°C:
-
If an alkene is protonated and the solvent is an alcohol rather than water, a reaction takes place that is very similar to acid-catalyzed hydration, but in the second step of the mechanism the...
-
Jenny wants to get good grades at school so she has decided to study more frequently. On any given day, the amount of time (in hours) that Jenny spends studying is normally distributed with mean 2.9...
-
McClellan Recreation manufactures and sells two models of paddle boards: Starter and Pro. The Starter model is a basic board used for instruction and purchased by novices. The Pro model is made with...
-
Computech Corporation is expanding rapidly and currently needs to retain all of its earnings; hence, it does not pay dividends. However, investors expect Computech to begin paying dividends,...

Study smarter with the SolutionInn App


