The following table presents the abundances and masses of the isotopes of zinc. What is the atomic
Question:
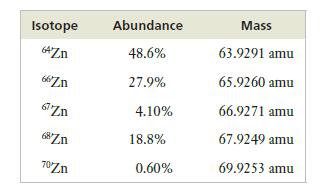
The following table presents the abundances and masses of the isotopes of zinc. What is the atomic weight of zinc?
Transcribed Image Text:
Isotope 64Zn 66Zn 67 Zn 68Zn 70Zn Abundance 48.6% 27.9% 4.10% 18.8% 0.60% Mass 63.9291 amu 65.9260 amu 66.9271 amu 67.9249 amu 69.9253 amu
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To calculate the atomic weight of zinc we need to use the ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
(A) The masses and percent isotopic abundances of the three naturally occurring isotopes of silicon are 28 Si, 27.9769265325 u, 92.223%; 29 Si, 28.976494700 u, 4.685%; 30 Si, 29.973377017 u, 3.092%....
-
Europium has two stable isotopes, 151 Eu and 153 Eu, with masses of 150.9197 u and 152.9212 u, respectively. Calculate the percent abundances of these isotopes of europium. EXAMPLE 2.2 Calculating...
-
Gallium has two naturally occurring isotopes, 69 Ga and 71 Ga, with masses of 68.9257 u and 70.9249 u, respectively. Calculate the percent abundances of these isotopes of gallium. EXAMPLE 2.2...
-
Lamonda Corp. uses a job order cost system. On April 1, the accounts had the following balances: The following transactions occurred during April: (a) Purchased materials on account at a cost of...
-
Which amortization method is required for intangibles? Are there any exceptions?
-
Terri Whelan, SS# 036-47-2189, is a Grade 1 Machinist who earns $20 per hour. On Monday June 12 through Wednesday June 14, she worked four hours per day on each of Jobs 007 and 2525. On Thursday and...
-
In 2013, Thomas DePrince, a passenger aboard a cruise ship, visited the ships jewelry boutique, operated by Starboard Cruise Services, Inc. DePrince told the employees of the boutique that he was...
-
Brockman Guitar Company is in the business of manufacturing top-quality, steel-string folk guitars. In recent years the company has experienced working capital problems resulting from the procurement...
-
2. What is the computational complexity of the following function Mystery1? int Mystery1(int n) { int sum = 0; for (int i=0; i
-
The atomic weight of copper is 63.55 amu. There are only two isotopes of copper, 63 Cu with a mass of 62.93 amu and 65 Cu with a mass of 64.93 amu. What is the percentage abundance of each of these...
-
Mercury is 16.716 times more massive than 12 C. What is the atomic weight of mercury? Remember to express your answer with the correct number of significant figures.
-
Rockwell Company owns a single restaurant which has a cantina primarily used to seat patrons while they wait on their tables. The company is considering eliminating the cantina and adding more dining...
-
To test the performance of its tires, a car travels along a perfectly flat (no banking) cir- cular track of radius 143 m. The car increases its speed at uniform rate of 5.07 m/s d|v| dt at = until...
-
Cup Movement (cm) 60 50 40 30 20 10 17 Condition 1 (15 cm) Rubber Band Stretch and Cup Movement 26 Condition 2 (30 cm) Rubber Band Stretch 53 Condition 3 (45 cm) 1. Do you see a trend in the points...
-
How many times (two, one, or none) does the apple pass through a point 1.80 m above your hand? If the apple does pass through this point, and is the apple moving upward or downward at each of these...
-
Objects with masses of 319 kg and 775 kg are separated by 0.441 m. A 46.1 kg mass is placed midway between them. 775 kg 319 kg 46.1 kg O -0.441 m Find the magnitude of the net gravitational force...
-
The bar graph on the right shows the average resistance to happiness, on a scale of 0 (no resistance) to 8 (completely resistant), for persons at various ages. The data in the graph can be modeled by...
-
Why are opportunity costs relevant when making decisions?
-
(a) Prove that form an orthonormal basis for R3 for the usual dot product. (b) Find the coordinates of v = (1, 1, 1)T relative to this basis. (c) Verify formula (5.5) in this particular case. 48-65...
-
Determine the configuration for every chirality center in each of the following compounds. a. b. c. HO H- - OH H,OH
-
Suggest an efficient synthesis for each of the following transformations: a. b.
-
Using acetylene as your only source of carbon atoms, identify a synthetic route for the production of 2-bromobutane.
-
Given the following demand and supply functions determine the equilibrium price and quantity Qd = 14-2P; Qs= 2+4P ii) What is the price elasticity of demand when the price is Sh. 4?
-
What would this accusation of discrimination cost the Bank of London? How should perceptions be managed inside and outside the organization? Gibb fully realized that policies that explicitly favoured...
-
Larkspur Service Corporation has 820,000 authorized shares of $20 par value common stock. As of June 30, 2025, there were 492,000 shares issued and outstanding. On June 30, 2025, the board of...

Study smarter with the SolutionInn App


