A mixture of hydrogen and nitrogen gas reacts as shown in the drawing below. (a) Write
Question:
A mixture of hydrogen and nitrogen gas reacts as shown in the drawing below.
(a) Write the balanced equation.
(b) Which reactant is the limiting reactant?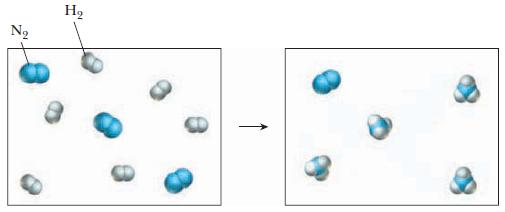
Transcribed Image Text:
N₂ H₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a 3H...View the full answer

Answered By

AKANKSHA CHATURVEDI
I am a organized professional with proven teaching guidance,and counseling skills.possess a strong track record in improving test scores and teaching effectively.I have ability to be a team player and resolve problems and conflicts professionally.Have an ability to communicate complex information in a simple and intertaining manner. Looking to contribute my knowledge and skills on platform like(solutionInn) that offers a genuine opportunity for career progression.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
A mixture of antimony atoms and Cl 2 in the gas phase reacts as shown in the drawing below. (a) Write the balanced equation. (b) Which reactant is the limiting reactant?
-
A mixture of hydrogen and uranium hexafluoride (UF6, molar mass: 352 kg/kmol) is stored in an abandoned oil well (in spherical chambers radioactive uranium can become critical leading to nuclear...
-
Lead(II) sulfide reacts with hydrogen peroxide to give lead(II) sulfate as shown in the unbalanced chemical equation PbS + H 2 O 2 PbSO 4 + H 2 O If 63.2 g of PbS is reacted with 48.0 g of H 2 O 2 ,...
-
The number of hours of daylight that occur at any location on Earth depends on the time of year and the latitude of the location. The equations below model the numbers of hours of daylight in Seward,...
-
A cotton reel is made up of a hub of radius a and two end caps of radius b. The mass of the complete reel is m and its moment of inertia about its longitudinal axis is I. The reel rests on a...
-
Graph the functions. The ts-plane (t-axis horizontal, s-axis vertical). What is the period of each function? What symmetries do the graphs have? S = CSC 2
-
For each of the following, decide whether there has been a valid offer: A . Would you like to buy my cabin in Maine? Its a beautiful piece of lakefront property and could sell for more than...
-
Thomas Kratzer is the purchasing manager for the headquarters of a large insurance company chain with a central inventory operation. Thomas's fastest-moving inventory item has a demand of 6,000 units...
-
1 Explain briefly about six sigma and capacity maturity models? 2 Explain about the contemporary management practices taking place in Indian business model? 3 4 What is performance management?...
-
1. Are the four intrinsic characteristics the best ones to base the relationship performance measures for Donnell Truong Ventures? If not, what characteristics would be more suitable? In either case,...
-
In a reaction of HCl and NaOH, the theoretical yield of H 2 O is 78.2 g. What is the theoretical yield of NaCl?
-
Lithium metal reacts with O 2 to form lithium oxide. What is the theoretical yield of lithium oxide when 0.45 g lithium reacts with excess O 2 ?
-
Indicate whether each of the following costs for a bicycle manufacturer is a product or a period cost, a variable or a fixed cost, a value-adding or a non-value adding cost, and, if it is a product...
-
Discuss the types of experimental observations that Lyon brought together in proposing her hypothesis concerning X-chromosome inactivation. In your own words, explain how these observations were...
-
In the clover butterfly, males are always yellow, but females can be yellow or white. In females, white is a dominant allele. Two yellow butterflies were crossed to yield an F1 generation consisting...
-
Three errors have been found in the records of T O Dorrigo and require correction by general journal. a The purchases account was used in error when a new photocopier (office equipment) was purchased...
-
In chickens, some varieties have feathered shanks (legs), but others do not. In a cross between a Black Langshan (feathered shanks) and a Buff Rock (unfeathered shanks), the shanks of the F1...
-
Lets suppose you were looking through a vial of fruit flies in your laboratory and noticed a male fly with pink eyes. What crosses would you make to determine if the pink allele is an X-linked gene?...
-
A monopolistically competitively firm faces the following demand and cost structure in the short-run. a. Complete the table b. What is the highest profit or lowest profit available to this firm? c....
-
2. Assume a person bends forward to lift a load "with his back" as shown in Figure P12.42a. The person's spine piv- ots mainly at the fifth lumbar vertebra, with the principal supporting force...
-
In Problem 7.4, we saw that an intramolecular substitution reaction can occur when the nucleophilic center and electrophilic center are present in the same compound. Draw the transition state of the...
-
Treatment of 5-hexen-1-ol with bromine affords a cyclic product: The mechanism of this reaction involves several steps, one of which is an intramolecular S N 2 process: In this step, a bond is in the...
-
Nicotine is an addictive compound found in tobacco, and choline is a compound involved in neurotransmission. The biosynthesis of each of these compounds involves the transfer of a methyl group from...
-
Q4 XYZ Ltd. has the following capital structure: Sources of Fund Equity Capital (400000 shares at INR 10) Ami (Rs.) 4000000 8% preference shares 10% Debentures Total 2000000 4000000 10000000 Market...
-
Doris Davis borrowed $8,200 on March 5 for 90 days at 8 3/4%. After 32 days, Doris made a payment on the loan of $2,700. On the 65th day, she made another payment of $2,500. What is her final payment...
-
Gardial GreenLights, a manufacturer of energy-efficient lighting solutions, has had such success with its new products that it is planning to substantially expand its manufacturing capacity with a...

Study smarter with the SolutionInn App


