Determine the rate constant and order by analyzing the concentration-time dependence. Reactant concentration (M) 0.6 0.5 0.4
Question:
Determine the rate constant and order by analyzing the concentration-time dependence.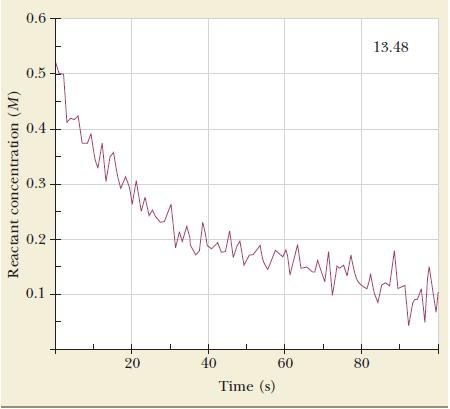
Transcribed Image Text:
Reactant concentration (M) 0.6 0.5 0.4 0.3 0.2 0.1 ww 20 win www 40 Time (s) 60 80 13.48
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
The integrated rate laws for various reaction orders can be used to calculate the rate constant and ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Explain the interrelation Between sociological theories of criminology (Strain,Anomie, social disorganization theory, cultural conflict, labeling theory, differential Association). With real examples.
-
In Example 15.3, you determined that the decomposition of SO 2 Cl 2 (under the given reaction conditions) is first order and has a rate constant of +2.90 * 10 -4 s -1 . If the reaction is carried out...
-
As in Example 7B.1, you are studying the decomposition of N 2 O 5 , but now you need to know how long it takes for a given amount of N 2 O 5 to decompose. A sample of N 2 O 5 is allowed to decompose...
-
For the following situations, comment on whether inspection by variables or by attributes might be more appropriate: a. Filling packaged food containers to the proper weight. b. Inspecting for...
-
The Lippmann-Schwinger formalism can also be applied to a one- dimensional transmission-reflection problem with a finite-range potential, V(x) ? 0 for 0 a. Suppose we have an incident wave coming...
-
1. In what phase of the venture life cycle is Jans firm currently operating? Defend your answer. 2. How are Jans actions helping to build an adaptive firm? Give three specific examples. 3. If Jans...
-
The following data were collected on specific gravity and spectrophotometer analysis for 26 mixtures of NG (nitroglycerine), TA (triacetin and 2 NDPA (2-nitrodiphenylamine). There is a need to...
-
On January 1, Intergen, Inc., invests $200,000 for a 40 percent interest in Ryan, a new joint venture with two other partners each investing $150,000 for 30 percent interests. Intergen plans to sell...
-
24. Write the standard form of the equation of the circle with the given characteristics. Endpoints of a diameter: (4,3), (-14, -13) 25. Write the equation of the circle in standard form, (x-h)+(y-k)...
-
When formic acid is heated, it decomposes to hydrogen and carbon dioxide in a first-order decay. At 550 C, the half-life of formic acid is 24.5 minutes. (a) What is the rate constant, and what are...
-
Nitrosyl chloride decomposes to nitrogen monoxide and chlorine at increased temperatures. Determine the rate constant and order from the concentration-time dependence. NOCI(g) NO(g) + Cl(g) Time (s)...
-
Assuming that 32 million histone octamers are required to package the human genome, how many histone molecules must be transported per second per nuclear pore complex in cells whose nuclei contain...
-
Mr. Taylor is a 52-year-old male with a medical history of episodic sinus bradycardia, which his General Practitioner (GP) states is due to his high level of fitness, occasional epigastric pain which...
-
If your capitalization rate is 12%, what is the value difference between a Net operating Income of $48,000 and $70,000?
-
I have chose a project working on a fast food hotel renovation. 1. Explain how EVM can be implemented in this individual project. 2. Explain how EVM will help your Individual Project. 3. What...
-
Analyze an organization's leadership effectiveness in up to five of James and Wooten's crisis phases, identifying leadership capacities, competencies, and behaviors (or lack thereof) in response to...
-
Write a function named naiveBayes. This function should get nxp feature matrix A of training data, nx1 label matrix C training data, 1xp feature matrix B of test sample, as inputs and return...
-
The comparative balance sheet of Fox Company, for the current year and the preceding year ended December 31, appears below in condensed form: current year preceding year Additional data for the...
-
A consumer magazine is evaluating five brands of trash compactors for their effectiveness in reducing the volume of typical household products that are discarded. In the experiment, each block...
-
Determine the moment at B, then draw the moment diagram for the beam. Assume the supports at A and C are rollers and B is a pin. EI is constant. 6 kN/m D B. -2 m 4 m 4 m
-
Determine the moments at B and C, then draw the moment diagram for the beam. Assume the supports at B and C are rollers and A is a pin. EI is constant. 300 Ib 200 lb/ft CE B -10 ft- 10 ft -8 ft-
-
Determine the moments at B and C, then draw the moment diagram for the beam. Assume the supports at B and C are rollers and A and D are pins. EI is constant. 12 kN/m 12 kN/m A 4 m 6 m 4 m
-
Why is it important not to judge people with the accent they have when they speak English ?
-
1. Define the unique characteristics of perfect competition. Does perfect competition exist in the real world? 2. How do monopolies maximize profits? 3. What are the unique characteristics of...
-
Describe the financing mechanism for providing health insurance in Italy - that is, what is the distribution/sources of funds used to provide health care coverage?

Study smarter with the SolutionInn App


