Given the thermochemical equations 2Cu(s) + Cl(g) 2CuCl(s) 2CuCl(s) + Cl(g) 2CuCl2(s) find the enthalpy change
Question:
Given the thermochemical equations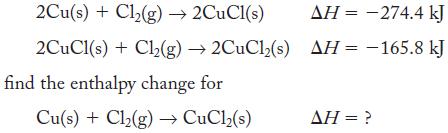
Transcribed Image Text:
2Cu(s) + Cl₂(g) → 2CuCl(s) 2CuCl(s) + Cl₂(g) →2CuCl2(s) find the enthalpy change for Cu(s) + Cl₂(g) → CuCl₂(s) AH = -274.4 kJ AH = -165.8 kJ AH = ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)

Answered By

BETHUEL RUTTO
Hi! I am a Journalism and Mass Communication graduate; I have written many academic essays, including argumentative essays, research papers, and literary analysis. I have also proofread and written reviews, summaries and analyses on already finished works. I am eager to continue writing!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
From the following data, calculate : (a) P/V Ratio. (b) Profit when sales are Rs. 40,000. (c) New break-even point if selling price is reduced by 20%. Fixed Expenses Rs. 8,000. Break-Even point Rs....
-
One way to evaluate fuels with respect to global warming is to determine how much heat they release during combustion relative to how much CO 2 they produce. The greater the heat relative to the...
-
Using the thermochemical equations in Exercise 5.67 as needed and in addition Exercise 5.67 Using the following thermochemical equations CH4(g) + 20(g) CO(g) + 2HO(l) CH4(g) + 30(g) 2CO(g) + 2HO(l)...
-
A strain of E-coli Beu 397-recA441 is placed into a nutrient broth at 30° Celsius and allowed to grow. The data shown in the table are collected. The population is measured in grams and the time...
-
For the shrinking-core model, if the rate of leaching is controlled by an interface chemical reaction that is first order in the concentration of reactant A, derive the expression, where k =...
-
Verify the formula differentiation. dx 1 V=sin- (4) + + C
-
Sofina Ali and Tristan Filippidis agreed to form a partnership by combining their businesses. The fair value and the carrying amount of the assets contributed by each partner and the liabilities...
-
For each of the items below, identify which fund would be used to account for the item and provide a justification for your answer. a. A city government issued general obligation bonds to finance the...
-
When an object is placed between the pole(P) and focus(F) of a concave mirror, the image formed is (i) behind the mirror (ii) virtual and erect and (iii) larger than the object (or magnified) M A' E...
-
Marider Industries makes two types of windbreaker jackets: one for spring and one for winter. Marider uses a traditional costing system and is considering switching to an Activity-Based Costing...
-
Draw an energy-level diagram that represents the Hesss law calculation in Exercise 5.71. Exercise 5.71 Given the thermochemical equations 2Cu(s) + Cl(g) 2CuCl(s) 2CuCl(s) + Cl(g) 2CuCl2(s) find the...
-
In the process of isolating iron from its ores, carbon monoxide reacts with iron(III) oxide, as described by the following equation: Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO(g) = -24.8 kJ The enthalpy change...
-
Briefly describe what an investment timing option is and why such options are valuable.
-
Revisit the guidelines in this chapter for negotiating a job offer. Then, with the two classmates you worked with for Exercise 16, practice negotiating. Divide roles: the hiring manager, candidate,...
-
An engine runs on a rich mixture of methyl and ethyl alcohol and air. At a pressure of 1 bar and \(10^{\circ} \mathrm{C}\) the fuel is completely vapourised. Calculate the air-fuel ratio by volume...
-
A gas engine with a volumetric compression ratio of \(10: 1\) is run on a weak mixture of methane \(\left(\mathrm{CH}_{4} ight)\) and air, with \(\phi=0.9\). If the initial temperature and pressure...
-
One method of reducing the maximum temperature in an engine is to run with a rich mixture. A spark-ignition engine with a compression ratio of 10:1, operating on the Otto cycle, runs on a rich...
-
A gas engine is operated on a stoichiometric mixture of methane \(\left(\mathrm{CH}_{4} ight)\) and air. At the end of the compression stroke, the pressure and temperature are \(10 \mathrm{bar}\) and...
-
True or false? a. All stocks in an equivalent-risk class are priced to offer the same expected rate of return. b. The value of a share equals the PV of future dividends per share.
-
Use the following data to answer the next two (2) questions: Product 1 Product 2 Product 3 Direct Material Cost $25,000 $30,000 $35,000 Direct Labor Cost $30,000 $40,000 $50,000 Direct Labor Hours...
-
Why is the inequality < 1 always satisfied in dilute electrolyte solutions?
-
Under what conditions does 1 for electrolyte solutions?
-
How do you expect S m for an ion in solution to change as the ionic radius increases at constant charge?
-
Retirement Assignment Tom and Jenny graduated college and got married. Both are in their 20s. Tom works for Accenture and earns $50,000 per year. Jenny works for Prudential and earns $40,000 per...
-
The Fraser Valley Soap company in British Columbia produces natural organic soaps. The company needs to purchase organic palm oil to make the natural soaps. The supplier of organic palm oil charges...
-
Figure 4 shows a position control of a wood crafting servo machine with a proportional derivative (PD) Controller. Given the parameters of Kp and Kd are 30 and 10 respectively. (a) For a non-delay...

Study smarter with the SolutionInn App


