Identify the hybrid orbitals used by antimony in SbCl 5 and in SbCl 6 , the
Question:
Identify the hybrid orbitals used by antimony in SbCl5 and in SbCl−6, the ion formed from the reaction of SbCl5 and Cl-. Explain your choices.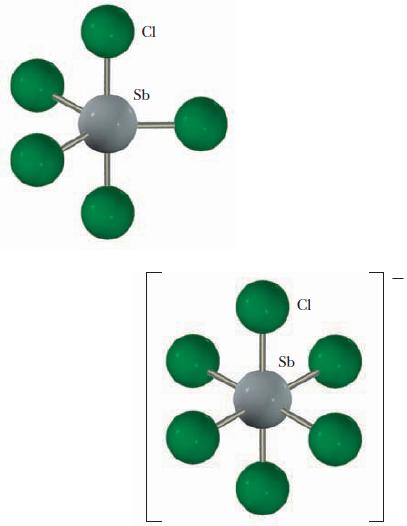
Transcribed Image Text:
CI Sb CI Sb
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The hybrid orbitals used by antimony Sb in the compounds SbCl and SbCl can be identified by consider...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Identify the hybrid orbitals used by boron in BCl3 and in BCl 4 , the ion formed from the reaction of BCl 3 and Cl - . Explain your choices.
-
Describe the hybrid orbitals used by each carbon atom in the following molecules: a. b. C-C-C-OH
-
Describe the hybrid orbitals used by each nitrogen atom in the following molecules: a. b. H-N-N-H
-
Prudence Corporation manufactures two products: X and Y. The company has 4,000 hours of machine time available and can sell no more than 800 units of product X. Other pertinent data follow. Which of...
-
Sustainable Growth Based on the following information calculates the sustainable growth rate for Hendrix Guitars, Inc.: Profit margin = 6.4% Total asset turnover = 1.70 Total debt ratio = .40 Payout...
-
What do the terms Europaper and Eurobonds refer to? Why are these instruments important to international banks and to their customers?
-
Comment on the statement: Classes provide the containers and objects provide the contents.
-
A firm implementing a diversification strategy has just acquired what it claims is a strategically related target firm but announces that it is not going to change this recently acquired firm in any...
-
At the end of the accounting period, Harris Company had $6,000 of par value common stock issued, additional paid in capital of $5,500, retained earnings of $6,000, and $2,000 of treasury stock. What...
-
Draw the energy-level diagram for the bonding and antibonding molecular orbitals for H 2 . Indicate their relative energies with respect to the 1s atomic orbitals of isolated hydrogen atoms.
-
Compare and contrast the molecular orbital and ionic bonding descriptions of LiF.
-
The summarised Balance Sheet of Twist Ltd is as follows. The preference shares had been issued at a premium of $0.20 per share. The preference shares are to be redeemed at $1.20 per share. No new...
-
Design a program that generates at random a series of 20 numbers in the range of 1 through 100. The program should store the numbers in a list and then display the following data: The lowest number...
-
Take this file as input: state_population.csv ( I cant upload the file so I took a picture of the numbers) Use the population field as the datapoint to select your sample. Set the margin of error...
-
Responsive to Direct Examination On cross examination, you will want to address issues that came up during the direct examination. You may gently lead your witness a bit more. You may also elicit...
-
For your discussion post: Complete the following. 1. Select the appropriate ICD-10 CM codes from Chapter 2 Neoplasms to code the scenario below. 2. Explain your rationale for choosing these codes....
-
Shell's purpose is to power progress together by providing more and cleaner energy solutions. Shell's strategy is to strengthen the position as a leading energy company by providing oil, gas and...
-
Judith loves cats, hates dogs, and is completely indifferent to tropical fish. Draw her indifference curves between (a) Cats and dogs, (b) Cats and fish, (c) Dogs and fish.
-
Before the 1973 oil embargo and subsequent increases in the price of crude oil, gasoline usage in the United States had grown at a seasonally adjusted rate of 0.57 percent per month, with a standard...
-
Determine the maximum normal stress developed in the bar when it is subjected to a tension of P = 2 kip. 0.125 in. 1.875 in. 1.25 in. r = 0.25 in. 0.75 in.
-
Determine the maximum axial force P that can be applied to the bar. The bar is made from steel and has an allowable stress of Ï allow = 21 ksi. 0.125 in. 1.875 in. 1.25 in. r = 0.25 in. 0.75 in.
-
The A-36 steel plate has a thickness of 12 mm. If Ï allow = 150 MPa, determine the maximum axial load P that it can support. Calculate its elongation, neglecting the effect of the fillets. r= 30...
-
How do you think the changes in the economic institution (for example: inflation, fewer jobs that pay a 'family' wage, the growing need for a college education) are affecting the social institution...
-
What is the significance to Canadian women of three of the trailblazers or women's organizations from my list that we have studied so far in CAN230? Add at least two reasons.
-
1) What are some of the social issues that writers for Television programs need to be extremely conscious of? 2) What is the social impact that the third screens have brought to people's lives? How...

Study smarter with the SolutionInn App


