In each part, identify the orbital diagram as the ground state, the excited state, or an impossible
Question:
In each part, identify the orbital diagram as the ground state, the excited state, or an impossible state. If it is an excited state, give the ground-state diagram, and if it is an impossible state, explain why.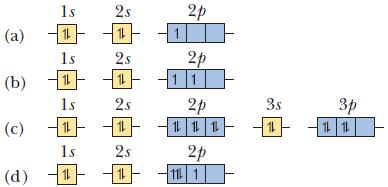
Transcribed Image Text:
(b) 15 (a) 14 (c) (d) 呻呻呻 15 11 15 14 15 1 2s 14 25 14 2s 11 25 11 1 20 2p 11 20 111 2p 11 1 3s 11 p 1L 1L
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a B ground state ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
The following orbital diagram represents an excited state of an atom. Identify the atom and give the orbital diagram corresponding to its ground state orbital diagram. [Ar] NNNNN 3d 4s 4p
-
Big Easy Business Patrick W. Malik and Jeffrey R. Sebree Shortly after Herb Loft man sat down in his office chair, his wife called, again complaining about some of their daughters wedding plans. This...
-
On January 1, 20x1, Allan Co. purchased 400,000 bonds for 392,000. The bonds mature on January 1, 20x5 and pay 12% annual interest beginning January 1, 20x2. Transaction costs are negligible. The...
-
Two particles of mass m 1 and m 2 are released from rest with infinite separation. Find their speeds v1 and v2 when their separation distance is r.
-
The cash data of Dunlap Automotive for July 2010 follow: Dunlap received the following bank statement on July 31, 2010: Additional data for the bank reconciliation include the following: a. The EFT...
-
Analyze the air pollution and mortality data in Table B. 15 using all possible regressions. Evaluate the subset models using the \(R_{p}^{2}, C_{p}\), and \(M S_{\text {Res }}\) criteria. Justify...
-
Siegel Corporation manufactures a product available in both a deluxe and a regular model. The company has made the regular model for years; the deluxe model was introduced several years ago to...
-
5. Here is the code (in Python) for the left rotation in an AVL tree: def rotate Left (root: Node) -> Node: Right LeftSubtree = Right Child.leftchild RightChild = root right child Right Child. left...
-
Annie opened a wholesale clothing business on 1 August 206. She received the following documents during August 206: Invoices August 4 From Sue, for goods, list price $1,960 less 25% trade discount 15...
-
An experiment uses single-photon counting techniques to measure light levels. If the wavelength of light emitted in an experiment is 589.0 nm, and the detector counts 1004 photons over a 10.0-second...
-
The distance between layers of atoms in a crystal is measured via diffraction of waves with a wavelength comparable with the distance separating the atoms. (a) What velocity must an electron have if...
-
Convert these decimal values to hex. (a)*75 (b) 314 (c)*2048 (d) 24 (e)*7245 (f) 498 (g)*25,619 (h) 4095 (i) 95 (j) 89 (k) 128 (l) 256
-
A thin piece of metal in the shape of a right triangle has inertia \(m\). Its perpendicular sides are of lengths \(\ell\) and \(2 \ell\). Determine the rotational inertia about an axis that runs (a)...
-
To see some of the trouble you can get into with noninertial reference frames, try this experiment. You and a friend stand opposite each other on the edge of a playground merry-go-round and get it...
-
Big Ben in London is the most accurate mechanical clock of its size. The \(300-\mathrm{kg}\) hour hand is \(2.7 \mathrm{~m}\) long, and the \(100-\mathrm{kg}\) minute hand is \(4.2 \mathrm{~m}\)...
-
Your aunt owns an amusement park, and she wants you to add a circular loop to an existing roller-coaster ride. The first hill for the existing roller coaster is \(55 \mathrm{~m}\) tall, and your aunt...
-
Hold a quarter down flat on a table with a finger. Then place a second quarter flat on the table, touching one point on the edge of the first quarter. If you roll the second quarter around the first...
-
In a study undertaken by Catalyst, 43% of women senior executives agreed or strongly agreed that a lack of role models was a barrier to their career development. In addition, 46% agreed or strongly...
-
Describe basic managerial approaches to implementing controls and how these are implemented.
-
Evaluate the vibrational partition function for NH 3 at 1000. K for which the vibrational frequencies are 950., 1627.5 (doubly degenerate), 3335, and 3414 cm 1 (doubly degenerate). Are there any...
-
Evaluate the vibrational partition function for CFCl 3 at 298 K, where the vibrational frequencies are (with degeneracy in parentheses) 1081, 847 (2), 535, 394 (2), 350., and 241(2) cm 1 .
-
Determine the populations in n = 0 and 1 for H 81 Br ( = 2649 cm 1 ) at 298 K.
-
A sail boat in the Great South Bay travels 2 miles east and then 4 miles south to get to Fire Island. What is its distance and displacement?
-
An air stripping tower is being designed to treat carbon tetrachloride from 500 to 5 g/L at a temperature of 25 C with a stripping factor of 8. Determine the operating airtowater ratio
-
If the wind force has a magnitude of 65 N , with what tension must the worker pull in order to move the box at a constant velocity? Express your answer with the appropriate units

Study smarter with the SolutionInn App


