Use Table 9.4 to calculate an approximate enthalpy change for (a) The combustion of 1 mol C
Question:
Use Table 9.4 to calculate an approximate enthalpy change for
(a) The combustion of 1 mol C2H4 in excess molecular oxygen to form gaseous water and CO2.
(b) The reaction of 1 mol formaldehyde, H2CO, with molecular hydrogen to form gaseous methanol (CH3OH).
Table 9.4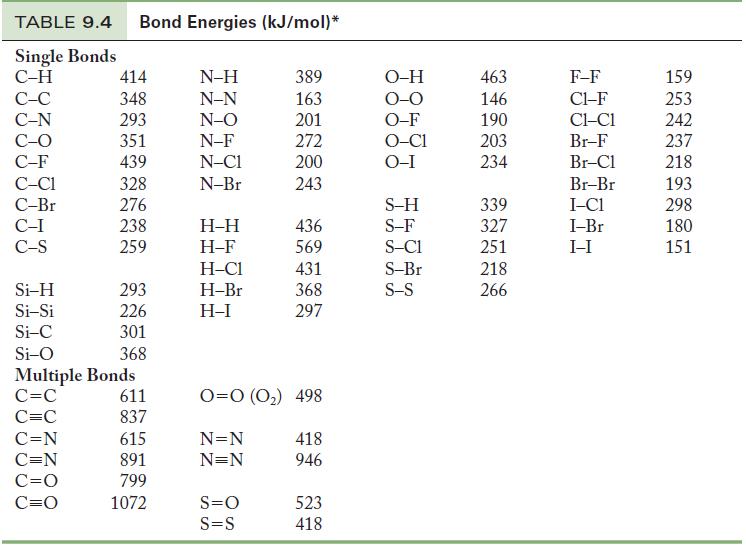
Transcribed Image Text:
TABLE 9.4 Bond Energies (kJ/mol)* Single Bonds C-H C-C C-N C-O C-F C-C1 C-Br C-I C-S Si-H Si-Si Si-C Si-O 414 348 293 351 439 328 276 238 259 C=N C=N C=O C=O 293 226 301 368 Multiple Bonds C=C C=C 611 837 615 891 799 1072 N-H N-N N-O N-F N-C1 N-Br H-H H-F H-C1 H-Br H-I 389 163 201 272 200 243 S=O S=S 436 569 431 368 297 0=0 (0₂) 498 N=N N=N 418 946 523 418 O-H O-O O-F O-C1 O-I S-H S-F S-C1 S-Br S-S 463 146 190 203 234 339 327 251 218 266 F-F C1-F CLC1 Br-F Br-Cl Br-Br I-C1 I-Br I-I 159 253 242 237 218 193 298 180 151
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a 1...View the full answer

Answered By

Sachin Singh
Dear Students,
I am Sachin, an M.Tech from IIT Delhi with several years of industrial experience in the software field. I am here to solve your problems, clear your doubts on that topic you find laborious, in the simplest of ways possible. I will make you understand the toughest of concepts with the easiest of approaches. You are going to find them very simple once we learn together.
I have spent years, have put in hundreds of hours in mastering my subjects. So, let me save you a lot of time understanding these subjects and solving the problems in an effortless fashion.
I hold a deep understanding of programming concepts, data structures, algorithms, digital electronics, discrete mathematics, etc.
I have varied experience in education and tutoring right from my schooling days. From the very start, I have been involved in teaching my fellow batch mates, juniors and kids around my neighborhood to help them with their assignments, complex problems and understanding any topic. People find my ways fun, engaging and interesting.
Teaching Style I follow :
Strong focus on the "why" in addition to the "what" while solving a problem or explaining a topic.
Root cause analysis for any problem/topic.
Easy examples to solve complex problems.
Strong commitment to clearing student's doubts until he/she completely understands it.
Friendly and compassionate teaching so that a student can express better.
I teach because I love sharing knowledge more than anything else in the world. In the end, Quoting this stirring quote(one of my favorites) by Swami Vivekananda that I follow :
Take up one idea. Make that one idea your life - think of it, dream of it, live on that idea. Let the brain, muscles, nerves, every part of your body, be full of that idea, and just leave every other idea alone. This is the way to success.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
List major external and internal customer groups of apple Explain the value exchange for each customer group listed of apple
-
Use Table 9.4 to calculate an approximate enthalpy change for (a) The reaction of H 2 and C 2 H 2 to form 1 mol C 2 H 6 . (b) The reaction of molecular hydrogen and molecular nitrogen to form 1 mol...
-
Using Table 9.4, calculate an approximate enthalpy change for (a) The reaction of molecular hydrogen (H 2 ) and molecular oxygen (O 2 ) in the gas phase to produce 2 mol water vapor. (b) The reaction...
-
Show the parse trees for the two parses that the grammar assigns for sentence S1. S1: the train station bus rumbles [3 marks] (b) Give an algorithm for a bottom-up passive chart parser without...
-
Calculate the radius of a vanadium atom, given that V has a BCC crystal structure, a density of 5.96 g/cm 3 , and an atomic weight of 50.9 g/mol.
-
Twin-Cities, Inc., purchased a building for $400,000. Straight-line depreciation was used for each of the first two years using the following assumptions: 25-year estimated useful life, with a...
-
Continuing Question 5, turn off the lag filter and use a notch filter to improve the performance. Simultaneously adjust Notch Freq and KVP to maximize KVP without inducing peaking in the closed-loop...
-
The Crunchy Granola Company is a diversified food company that specializes in all natural foods. The company has three operating divisions organized as investment centers. Condensed data taken from...
-
Rose Hill, a soybean farm in northern Minnesota, has a herd of 4 5 dairy cows. The cows produce approximately 2 , 5 2 0 gallons of milk per week. The farm currently sells all its milk to a nearby...
-
The equation for the combustion of gaseous methanol is Using the bond dissociation enthalpies, estimate the enthalpy change for this reaction. 2CH3OH(g) + 302(g) 2CO(g) + 4HO(g)
-
Use the octet rule to predict the element (E) from the second period that would be the central atom in the following ions. (a) EF (b) EF+
-
What is Quantity Demanded and how it effects the economies?
-
1) Consider a wave packet for which A(K) = {0 (N, Obtain (x, 0), Ak, Ax and show that Ak^x is independent of k - 2K x 2K elsewhere
-
A 0.450 kg block rests on a frictionless horizontal surface, where it is attached to a massless spring whose k-value equals 18.0 N/m. Let x be the displacement, where x = O is the equilibrium...
-
Two donkeys are tied to the same pole. One donkey pulls the pole at a strength of 6 N. The other donkey pulls the pole at a strength of 3.5 N in a direction that is 80 counterclockwise from the...
-
(d) Indicate the SI units for the dimensions Q0 L in derived form Which SI units can divide these units to give the dimensions of specific heat (which is QM 0-? What is this quantity called?
-
Using the standard thermodynamic data, calculate the Ksp at 298K for NaBr. AG for Br -104.0 kJ/mol AG for Na-261.90 kJ/mol AG:for NaBr = -349.0 kJ/mol NaBr (3) Na (aq) + Br (aq)
-
Does stagflation contradict the theory of the Phillips curve?
-
Define cultural intelligence. Cite the books or journal articles you found in Capella's library. Explain why cultural intelligence is important for HR practitioners and other organizational managers.
-
The rods each have the same 25-mm diameter and 600-mm length. If they are made of A992 steel, determine the forces developed in each rod when the temperature increases by 50° C. -600 mm 60 /60...
-
Determine the resultant internal loadings on the cross section at point D. 1.25 kN/m 1.5 m '0.5 m' 0.5 m'0.5 m
-
Determine the resultant internal loadings at cross sections at points E and F on the assembly. 1.25 kN/m 1.5 m '0.5 m'0.5 m'0.5 m
-
Design a flowchart for finding the product of numbers 1 through 100. Thus, output = 1 2 3 4 5 6 ....... 99 100. Find a scalable solution, i.e. if you had to multiply numbers from 1 to 100000, your...
-
Given your answers to questions 3 and 4, we want to call the customers who have priorities that are Medium or higher (Medium, High or Critical) but do not choose Express Air for delivery. Populate...
-
You are the AS3 in the TOC when a nearby UAS reports seeing a team emplacing rockets towards the base. You call to the Apache Troop to have them launch with the forces at hand and attack based on...

Study smarter with the SolutionInn App


