Use Table 9.4 to calculate an approximate enthalpy change for (a) The reaction of H 2 and
Question:
Use Table 9.4 to calculate an approximate enthalpy change for
(a) The reaction of H2 and C2H2 to form 1 mol C2H6.
(b) The reaction of molecular hydrogen and molecular nitrogen to form 1 mol ammonia.
Table 9.4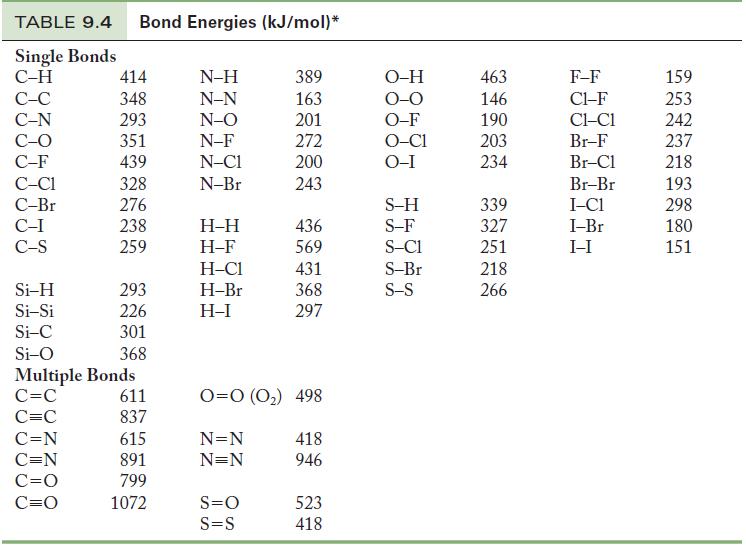
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a HCCH 2HH CH3CH3 dissociation bonds broken EAH bonds formed H ...View the full answer

Answered By

Robert Mbae
I have been a professional custom essay writer for the last three years. Over that period of time, I have come to learn the value of focusing on the needs of the clients above everything else. With this knowledge, I have worked hard to become an acclaimed writer that can be trusted by the customers to handle the most important custom essays. I have the necessary educational background to handle projects up to the Ph.D. level. Among the types of projects that I've done, I can handle everything within Dissertations, Project Proposals, Research Papers, Term Papers, Essays, Annotated Bibliographies, and Literature Reviews, among others.
Concerning academic integrity, I assure you that you will receive my full and undivided attention through to the completion of every essay writing task. Additionally, I am able and willing to produce 100% custom writings with a guarantee of 0% plagiarism. With my substantial experience, I am conversant with all citation styles ranging from APA, MLA, Harvard, Chicago-Turabian, and their corresponding formatting. With all this in mind, I take it as my obligation to read and understand your instructions, which reflect on the quality of work that I deliver. In my paper writing services, I give value to every single essay order. Besides, whenever I agree to do your order, it means that I have read and reread your instructions and ensured that I have understood and interpreted them accordingly.
Communication is an essential part of a healthy working relationship. Therefore, I ensure that I provide the client with drafts way long before the deadline so that the customer can review the paper and comment. Upon completion of the paper writing service, the client has the time and right to review it and request any adjustments before releasing the payment.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Use Table 9.4 to calculate an approximate enthalpy change for (a) The combustion of 1 mol C 2 H 4 in excess molecular oxygen to form gaseous water and CO 2 . (b) The reaction of 1 mol formaldehyde, H...
-
Using Table 9.4, calculate an approximate enthalpy change for (a) The reaction of molecular hydrogen (H 2 ) and molecular oxygen (O 2 ) in the gas phase to produce 2 mol water vapor. (b) The reaction...
-
List major external and internal customer groups of apple Explain the value exchange for each customer group listed of apple
-
Discuss why marketing management is important to an organization, and how corporate and business strategy analysis gives an organization a competitive advantage.
-
Calculate the radius of an iridium atom, given that Ir has an FCC crystal structure, a density of 22.4 g/cm 3 , and an atomic weight of 192.2 g/mol.
-
The following data pertain to three divisions of Calrisian Enterprises. The companys required rate of return on invested capital is 8 percent. Required: Fill in the blanksabove. Division I Division...
-
Provide the ideal gain scheduling for Figure 12.16 as a function of Q. DATA FROM FIGURE 12.16 Assume inertia varies with theta as follows: where JM is the motor inertia, JL is the load inertia, and R...
-
For the following situations, would you collect information using a sample or a population? Why? a. Statistics 201 is a course taught at a university. Professor Rauch has taught nearly 1,500 students...
-
What strategies are effective for managing resistance to change, particularly in the context of entrenched organizational cultures and power dynamics ?
-
The equation for the combustion of gaseous methanol is Using the bond dissociation enthalpies, estimate the enthalpy change for this reaction. 2CH3OH(g) + 302(g) 2CO(g) + 4HO(g)
-
Use the octet rule to predict the element (E) from the second period that would be the central atom in the following ions. (a) EF (b) EF+
-
Water is boiling at 1 atm pressure in a stainless steel pan on an electric range. It is observed that 1.25 kg of liquid water evaporates in 30 min. The rate of heat transfer to the water is (a) 1.57...
-
Which of the subshell designations are possible and which are impossible? 2f 4d Possible 2p 1.5p Answer Bank ld Impossible
-
A sample of CH 4 contains 6.6510 22 molecules. How many moles of CH 4 are in this sample? How many moles of hydrogen atoms are in this sample?
-
The vapor pressure, P, of a certain liquid was measured at two temperatures, T. The data is shown in the table. T(K) P (kPa) 225 3.31 775 8.38 Keep the pressure units in kilopascals. If you were...
-
Pick up a token and place it on the screen. If you're stuck, ask yourself these questions: 1. Are there electronegativity differences in the molecule? 2. How is the molecule's geometry contributing...
-
3. Determine the K and pK (to two decimal places) for an aqueous solution containing 0.19 M of a weak, monoprotic acid, HA, with a pH of 5.10. HA(aq) + HO(1) H3O+ (aq) + A(aq) = 0 225 qrimen A: 3.32x...
-
Why do economists who believe people form rational expectations have little faith that announced changes in monetary policy will have substantial effects on real output?
-
During 2012, Cheng Book Store paid $483,000 for land and built a store in Georgetown. Prior to construction, the city of Georgetown charged Cheng $1,300 for a building permit, which Cheng paid. Cheng...
-
The beam supports the distributed load shown. Determine the resultant internal loadings acting on the cross section at point C. Assume the reactions at the supports A and B are vertical. 4 kN/m A...
-
The beam supports the distributed load shown. Determine the resultant internal loadings acting on the cross section at point D. Assume the reactions at the supports A and B are vertical. 4 kN/m -1.5...
-
Determine the resultant internal loadings acting on the cross sections at points D and E of the frame. 4 ft 75 Ib/ft LA B. 2 ft 2 ft 1 ft 1 ft 30 1 ft 150 lb
-
Give a Literature review on "How education IT administrators prevent ransomware attacks in an open-access environment" ?
-
Describe four key advantages of Virtualization. Explain why type 1 hypervisor should be used in a server environment, such as data center.
-
Using the link http://learninga-z.com explore the Differentiated Instructional Strategies document. Describe a strategy that is interesting

Study smarter with the SolutionInn App


