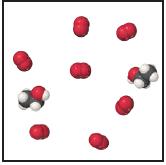
Consider the mixture of ethanol, C 2 H 5 OH, and O 2 shown in the accompanying
Question:
Consider the mixture of ethanol, C2H5OH, and O2 shown in the accompanying diagram.
(a) Write a balanced equation for the combustion reaction that occurs between ethanol and oxygen.
(b) Which reactant is the limiting reactant?
(c) How many molecules of CO2, H2O, C2H5OH, and O2 will be present if the reaction goes to completion?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a The given combustion reaction between ethanol and oxygen C 2 H 5 OHl O 2 g CO 2 g H 2 Og Let us ba...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Ethyl alcohol (ethanol) can be produced by the fermentation of sugars derived from trains and other agricultural products. Some countries without large petroleum and natural as reservessuch as...
-
Ethyl alcohol can be bacterially oxidized to acetic acid in the following two-step fermentation sequence: 2 C2H5OH + O2 2 CH3CHO + 2 H2O 2 CH3CHO + O2 2 CH3COOH If the alcohol-containing feedstock...
-
Nitrogen monoxide and oxygen react to form nitrogen dioxide. Consider the mixture of NO and O2 shown in the accompanying diagram. The blue spheres represent N, and the red ones represent O. (a) Draw...
-
In this photo, Ramu is eating raw meat. What do Ramus behaviors suggest about how we develop self-concepts? Would you define Ramu as a human or a wolf?
-
Solve the system of equations using Gaussian elimination or Gauss-Jordan elimination. [6.3] a. x + 2y = 5, 2x - 5y = -8 b. 3x + 4y + 2z = 3, 5x - 2y - 13z = 3, 4x + 3y - 3z = 6
-
Question 1 Consider three assets: two risky assets (asset 1 and asset 2) and the riskless asset. Asset 1 has an expected return of 5% and a volatility of 10%. Asset 2 has an expected return of 10%...
-
Consider the simple linear regression model for the baseball data using Team ERA as the predictor. Find the value of the PRESS statistic and the $R^{2}$ based on PRESS for this model. What...
-
1. Identify any potential threats to judgment you think could exist based on the facts of the case? 2. Thinking back to the biases discussed in Chapter 2, what biases might the identified threat(s)...
-
A company is projected to generate free cash flows of $429 million next year, growing at a 4.7% rate until the end of year 3. After that, cash flows are expected to grow at a stable rate of 2.6%. The...
-
Flowmaster Forge Inc. is a designer and manufacturer of industrial air-handling equipment that is a wholly owned subsidiary of Howden Industrial Inc. Howden is interested in selling Flowmaster to an...
-
If Avogadros number of pennies is divided equally among the 314 million men, women, and children in the United States, how many dollars would each receive? How does this compare with the gross...
-
Consider the mixture of propane, C 3 H 8 , and O 2 shown below. (a) Write a balanced equation for the combustion reaction that occurs between propane and oxygen. (b) Which reactant is the limiting...
-
Under what conditions will a transfer of receivables be recognized as a sale/derecognition? As a borrowing? What financial statement ratios are particularly affected by these alternatives?
-
Assume that an investment in the S&P 5 0 0 index gives an average return of 1 1 % . The short - term US government bond provides a risk - free return of 3 . 5 % . a . If you aim to earn an expected...
-
The stock of LightSpace Inc. was expected to pay a $3 dividend last year and had an anticipated growth rate of 7%. The stock was selling for $26 at the time. What would be the expected rate of return...
-
The Family and Medical Leave Act (FMLA) permits eligible employees which of the following? up to 12 work weeks of paid leave during any 12-month period up to 10 work weeks of paid leave during any...
-
The table below contains the covariance matrix of stock returns and the market. Assume that the assumptions of CAPM hold "Harket" 0.1512 "Market" "BlueChip" "YellowChip" "BlackChip""WhiteChip" 0.0969...
-
A firm with $ 2 0 book equity value per share, and 1 2 % required rate of return from shareholders, intends to maintain a constant dividend payout ratio of 6 0 % and a constant ROE of 1 0 % in the...
-
The time taken by a randomly selected applicant for a mortgage to fill out a certain form has a normal distribution with mean value 10 min and standard deviation 2 min. If five individuals fill out a...
-
Highland Theatre is owned by Finnean Ferguson. At June 30, 2014, the ledger showed the following: Cash, $6,000; Land, $100,000; Buildings, $80,000; Equipment, $25,000; Accounts Payable, $5,000;...
-
(a) What is a hydrocarbon? (b) Butane is the alkane with a chain of four carbon atoms. Write a structural formula for this compound and determine its molecular and empirical formulas.
-
(a) What ending is used for the names of alkanes? (b) Hexane is an alkane whose structural formula has all its carbon atoms in a straight chain. Draw the structural formula for this compound and...
-
(a) What is a functional group? (b) What functional group characterizes an alcohol? (c) With reference to Exercise 2.75, write a structural formula for 1-butanol, the alcohol derived from butane, by...
-
Compare and contrast strategic leadership against other leadership models including situations that call for strategic leadership versus other models of leadership and why.
-
Describe the role of Leadership in healthcare. Describe the role of Strategic Leadership in healthcare. Describe the difference between Leadership and Strategic Leadership in healthcare.
-
What is a good Annotated Bibliography for the following three books? Re-Thinking Strategic Leadership in the Digital Age by Sabine Dembkowski Exploring the Concept of Strategic Leadership by...

Study smarter with the SolutionInn App


