An ideal monatomic gas is taken through the cycle in the PV diagram. (a) If there are
Question:
An ideal monatomic gas is taken through the cycle in the PV diagram.
(a) If there are 0.0200 mol of this gas, what are the temperature and pressure at point C?
(b) What is the change in internal energy of the gas as it is taken from A to B?
(c) How much work is done on this gas per cycle?
(d) What is the total change in internal energy of this gas in one cycle?
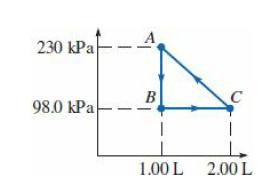
Transcribed Image Text:
230 КPа B 98.0 КРа 1.00 L 2.00 L
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a The pressure at point C from the graph is P 98 kPa 98 10 3 Pa Appl...View the full answer

Answered By

Umair Expert
Hi Everyone.
I have 6 years of teaching experience.
I am serving as a tutor 2 more websites.
I will provide you projects and solutions of questions related to any subject.
I am a good programmer.(PHP, Python)
I am good command in mathematics.
I am here to assist you.
I'll be very happy to work with you...
Thanks dear..
0.00
0 Reviews
10+ Question Solved
Related Book For 

College Physics With An Integrated Approach To Forces And Kinematics
ISBN: 978-1260547719
5th Edition
Authors: Alan Giambattista
Question Posted:
Students also viewed these Sciences questions
-
An ideal monatomic gas is taken through the cycle in the PV diagram. (a) If there are 0.0200 mol of this gas, what are the temperature and pressure at point C? (b) What is the change in internal...
-
A 1.00-mol sample of an ideal monatomic gas is taken through the cycle shown in Figure P22.62. The process A B is a reversible isothermal expansion. Calculate (a) the net work done by the gas, (b)...
-
If the pressure of an ideal monatomic gas is increased while the number of moles is kept constant, what happens to the average translational kinetic energy of one atom of the gas? Is it possible to...
-
The table represents values of differentiable functions f and g and their first derivatives. Use the table of values to answer the questions that follow. Work all of the parts below the line. X f g...
-
McAfee FurniturereceivedaninvoicedatedSeptember14fromPalisadeManufacturing with terms 2 /10, EOM for the following items: four Prestige bedroom suites at $3900 each less 20%, 7%, and six Oak...
-
Your new co-worker just came in and made the following statement: 'Every Friday is casual day around here; people wear casual clothes to work on Fridays. Jan, over there, is wearing jeans and a...
-
For the linear part of the moisture absorption curve for a temperature of \(77^{\circ} \mathrm{C}\) in Figure 5.12, and assuming a specimen thickness of \(2.54 \mathrm{~mm}\), use the relevant...
-
A house at the bottom of a hill is fed by a full tank of water 5.0 m deep and connected to the house by a pipe that is 110m long at an angle of 58o from the horizontal (Fig. 10-50). (a) Determine the...
-
StorSmart Company makes plastic organizing bins. The company has the following inventory balances at the beginning and end of March: S Raw materials Work in process Finished goods Beginning Inventory...
-
At the end of 2020, Carpenter Co. has accounts receivable of $700,000 and an allowance for doubtful accounts of $54,000. On January 24, 2021, the company learns that its receivable from Megan Gray is...
-
As an ideal gas is compressed at constant temperature, (a) Heat flows out of the gas. (b) The internal energy of the gas does not change. (c) The work done on the gas is zero. (d) None of the above...
-
An ideal gas is confined to the left chamber of an insulated container. The right chamber is evacuated. A valve is opened between the chambers, allowing gas to flow into the right chamber. After...
-
Use a browser to search for child labor. How extensive is the problem of child labor? How do you think child labor affects the childs daily life? How does it affect the childs future? What about the...
-
Can i please have some clarification on if these journals are correct? Trial balance and adjustments are attached for reference. i. ii. iii. iv. V. vi. vii. Adjust sales revenue for $1,600 that was...
-
Submit quiz During its first year of operations, Precious Gems Collection Inc. had sales of $1,010,000, all on account. Industry experience suggests that Precious Gems Collection's bad debt expense...
-
2. (3+6+2 points) Expected utility theory You are risk neutral with [U(x) = x], and care only about your income in the current period. With probability p, you will catch a disease that reduces your...
-
Red blood cells can often be charged. Consider two red blood cells with the following charges: 1 6 . 6 pCand + 4 8 . 6 pC . The red blood cells are 3 . 7 0 cm apart. What is the magnitude of the...
-
Selected T-accounts of Moore Company are given below for the just completed year: Raw Materials Manufacturing Overhead Debit Credit Debit Credit Balance 1/1 Debits 20,000 Credits 130,000 ? Debits...
-
Evaluate (a) (cx2y ds using the parameterization x = 3 sin t, y = 3 cos t, 0 ( t ( (/2, which reverses and orientation of C I Example 1, and (b) (c4xy2 dx + xy2 by using the parameterization x = 3 -...
-
How do individual companies respond to economic forces throughout the globe? One way to explore this is to see how well rates of return for stock of individual companies can be explained by stock...
-
A spaceship travels at constant velocity from Earth to a point 710 ly away as measured in Earth's rest frame. The ship's speed relative to Earth is 0.9999c. A passenger is 20 yr old when departing...
-
Describe the photoelectric effect and four aspects of the experimental results that were puzzling to nineteenth century physicists. How does the photon model of light explain the experimental results...
-
If green light causes the ejection of electrons from a metal in a photoelectric effect experiment and yellow light does not, what would you expect to happen if red light were used to illuminate the...
-
Assume that management wants wage concession from its counterpart labor union. Explain why this often leads to disputes. Suppose management gives up some of its future negotiating power by subjecting...
-
Why have you chosen this nursing Field of Specialization or Major? What influenced you in selecting your intended major of study and what goal(s) have you set for the future?
-
What are logical fallacies and how can they affect our decision-making?

Study smarter with the SolutionInn App


