Estimate whether entropy increases or deceases with the following reaction. Use data from Table 17.1 to confirm
Question:
Estimate whether entropy increases or deceases with the following reaction. Use data from Table 17.1 to confirm your estimate.
2 C(s) + 3 H2(g) → C2H6(g)
Table 17.1
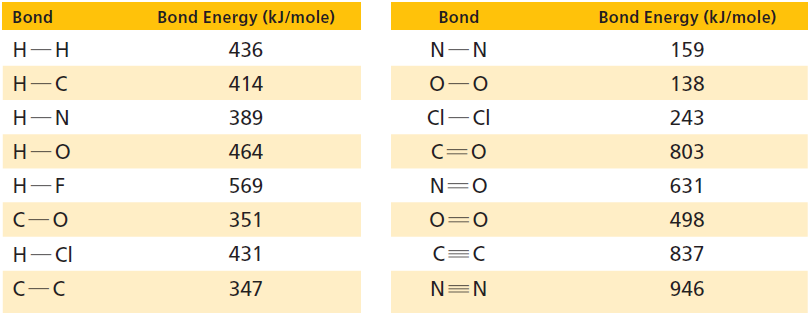
Transcribed Image Text:
Bond Energy (kJ/mole) Bond Energy (kJ/mole) Bond Bond Н-Н 436 159 Н-С 414 138 Cl-CI 389 Н—N 243 803 Н—О 464 C=0 Н-—F 569 N=0 631 351 498 Н— СІ 431 837 N=N 347 946
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (9 reviews)
Consider how this reaction shows the formation of a single molec...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
Estimation of diffusivity for a binary mixture at high density, Predict CD AB for an equimolar mixture of N 2 and C 2 H 6 at 288.2K and 40atm (a) Use the value of D AB at 1atm from Table 17.1-1,...
-
The rates of many atmospheric reactions are accelerated by the absorption of light by one of the reactants. For example, consider the reaction between methane and chlorine to produce methyl chloride...
-
How many grams of gallium are there in a 145-gram sample of gallium arenside, GaAs?
-
Find the magnitude of the reaction at A and the tension in cable CD using (a) the con- cepts of three-force bodies (closed polygon method discussed in chapter 2). (b) Check your answers by doing sum...
-
Kane Legal Services provides legal advice to clients. The following data apply to the first six months of operation: Required a. What is the average service revenue per hour for the six-month time...
-
As in Question 5, you have been asked to quantify the effects of removing an import duty; somebody has already estimated the effects on the countrys production, consumption, and imports. This time...
-
A laminar boundary layer velocity profile is approximated by \(u / U=2(y / \delta)-2(y / \delta)^{3}+(y / \delta)^{4}\) for \(y \leq \delta\), and \(u=U\) for \(y>\delta\). (a) Show that this profile...
-
Rice Company performed services on account for $30,000 in 2013, its first year of operations. Rice collected $24,000 cash from accounts receivable during 2013 and the remaining $6,000 in cash during...
-
A diver of mass 63.3 kg stands on one end of a diving board of mass 35.9 kg, as shown in the diagram. Everything is in equilibrium (since nothing is moving). What is the magnitude of the support...
-
You are a staff accountant at Anderson & Paul, a CPA firm located in downtown San Diego. A long-time client, Brady Patterson, has contacted you with a request. Brady is 32 years old, is beginning...
-
According to the second law of thermodynamics, exothermic reactions, such as the burning of wood, are favored because they result in the dispersal of energy. Wood, however, will not spontaneously...
-
Exothermic reactions are favored because they release heat to the environment. Would an exothermic reaction be more favored or less favored if it were carried out within a superheated chamber?
-
Sketch the general appearance of the curve for the titration of a weak base with a strong acid. Explain (in words) what chemistry governs the pH in each of the four distinct regions of the curve.
-
What are time-series data? Give three examples of key macroeconomic variables that are examples of time-series data.
-
Assume that a period of deflation leads to a rise in the supply of real money balances. Explain the effect of this change on the economy using the ISLM model and then what effect it would have on AD...
-
Distinguish between planned expenditure and actual expenditure.
-
What does the IS curve show? What does the LM curve show?
-
Germany generally runs a trade surplus. Do you think this is most related to high foreign demand for German goods, low German demand for foreign goods, a high German saving rate relative to German...
-
An increase in the government budget deficit _______. A. increases private saving and investment B. increases private saving and decreases investment C. increases the supply of private saving and...
-
Ann hires a nanny to watch her two children while she works at a local hospital. She pays the 19 year-old nanny $125 per week for 48 weeks during the current year. a. What is the employers portion of...
-
Consider the circuit shown in Fig. P25.73. The emf source has negligible internal resistance. The resistors have resistances R 1 = 6.00 Ω and R 2 = 4.00 Ω. The capacitor has...
-
Consider the circuit shown in Fig. P25.72. The battery has emf 72.0 V and negligible internal resistance. R 2 = 2.00 Ω, C 1 = 3.00 µF, and C 2 = 6.00 µF. After the capacitors...
-
Compact fluorescent bulbs are much more efficient at producing light than are ordinary incandescent bulbs. They initially cost much more, but they last far longer and use much less electricity....
-
Shown are selected financial data for Another World and Imports, Inc., at the end of the current year. Net credit sales Cost of goods sold Cash Another World Imports, Inc. $ 675,000 $560,000 504,000...
-
The following partially complete T-accounts for the month of June along with additional information are from Renfrew & Company: Materials Inventory Work-in-Process Inventory Finished Goods Inventory...
-
The information that follows pertains to Julia Company: (a) Temporary differences for the year 2024 are summarized below. Expenses deducted in the tax return, but not included in the income...

Study smarter with the SolutionInn App


