Rank the scale readings from greatest to least. (Ignore friction.) B -? IG-B
Question:
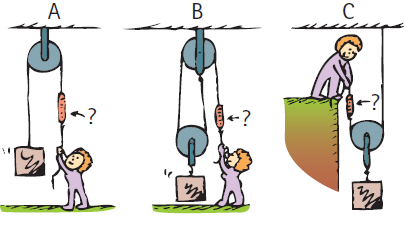
Transcribed Image Text:
B -? IG-B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
A...View the full answer

Answered By

Sandip Nandnawar
I am a B.E (Information technology) from GECA and also have an M.C.M from The University of RTMNU, MH.
I worked as a software developer (Programmer and TL). Also working as an expert for the last 6 years and deal with complex assessment and projects. I have a team and lead a team of experts and conducted primary and secondary research. I am a senior software engg and senior expert and deal with all types of CSE and IT and other IT-related assessments and projects and homework.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
All bulbs are identical in the following circuits. A voltmeter is connected across a single bulb to measure the voltage drop across it. Rank the voltage readings from greatest to least.
-
From greatest to least, rank the frequency of radiation of these emitters of radiant energy: (a) Red-hot star. (b) Blue-hot star. (c) Sun.
-
Different materials, A, B, C, and D, rest on a table. a. From greatest to least, rank them by how much they resist being set into motion. b. From greatest to least, rank them by the support (normal)...
-
In year 1, the prices of goods X, Y, and Z are $2, $4, and $6 per unit, respectively. In year 2, the prices of good X, Y, and Z are $3, $4, and $7, respectively. In year 2, twice as many units of...
-
Determine the mass defect (in atomic mass units) for (a) helium , which has an atomic mass of 3.016030 u, and (b) , which has an atomic mass of 3.016050 u. (c) On the basis of your answers to parts...
-
Define (a) Millimole. (b) Titration. (c) Stoichiometric ratio. (d) Titration error.
-
Why does a use case need a unique, numeric identifier?
-
MULTIPLE CHOICE QUESTIONS 1. Which of the following statements best explains why the CPA profession has found it essential to promulgate ethical standards and to establish means for ensuring their...
-
The following information about Sung Company on January 1, 2014 was available: Book Value Fair Value: Inventories 20,000 28,200 Building 60,000 87,000 Total 80,000 115,200 Accounts Payable 1,000...
-
The Cinnamon case: Sales negotiations (Role Play) - (B) The buyer You are the owner of Offshoot Intermediaries Limited, a family-run enterprise that offers various drug formulations and baby food...
-
Starting from rest, Megan zooms down a frictionless slide from an initial height of 4.0 m. Show that her speed at the bottom of the slide is 80 m/s, or 8.9 m/s.
-
A lunar vehicle is tested on Earth at a speed of 12 km/h. When it travels at that same speed on the Moon, is its momentum more, less, or the same?
-
The U.S. government's flagship international educational exchange program, the Fulbright program, is sponsored by the Bureau of Educational and Cultural Affairs. Fulbrights are awarded based on...
-
(7.8.c] What is output when the following code is executed? def foo(list): for i in range(len(list)): list[i] = list[i] 2 def main(): 1st 13, 7, 4, 5, 8, 10] foo (1st) print (1st) main()
-
Packer, Inc., a U.S. producer of computer disks, plans to establish a subsidiary in Mexico in order to penetrate the Mexican market. Packer's executives believe that the Mexican peso's value is...
-
Silicon Valley Bank has recently published their financial results for FY2023. The Company managed to generate $1.4 billion (bn) in sales with a 30% gross margin and a 5% net margin. Depreciation for...
-
What are the advantages and disadvantages of each main type of life insurance (term life insurance, whole life insurance, variable life insurance, universal life insurance and annuities) ? What are...
-
A firm has $10m ($1m shares @ $10 each). The firm is planning to invest this $10m in a year-long project that will generate $14m after one year. If the opportunity rate is 13%, find NPV of the...
-
Draw the structure of the compound with molecular formula C8H11N that exhibits the following 1 H NMR and 13 C NMR spectra: Proton NMR 2 22 Chemical Shift (ppm) Carbon NMR 128.8 128.4 40.0 -126.1...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
Conventional hot-water heaters consist of a tank of water maintained at a fixed temperature. The hot water is to be used when needed. The drawbacks are that energy is wasted because the tank loses...
-
A 15.0-g bullet traveling horizontally at 865 m/s passes through a tank containing 13.5 kg of water and emerges with a speed of 534 m/s. What is the maximum temperature increase that the water could...
-
One suggested treatment for a person who has suffered a stroke is immersion in an ice-water bath at 0C to lower the body temperature, which prevents damage to the brain. In one set of tests, patients...
-
Silverton Confectionery is a growing Berkshire-based company specialising in selling quality chocolates and sweets at higher than average prices through newsagents and confectioners. At present their...
-
4. X, the proprietor of a departmental store, decided to calculate separate profits for his two departments L and M for the month ending 31st January. Stock on 31st January could not be valued for...
-
What level of confidentiality should be attached to the preparation and handling of a memorandum of law? Why? Assume you have been working for a legal specialist in estate law for a number of years...

Study smarter with the SolutionInn App


