Draw the structure of the compound with molecular formula C8H11N that exhibits the following 1 H NMR
Question:
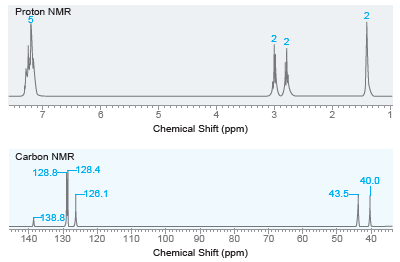
Transcribed Image Text:
Proton NMR 2 22 Chemical Shift (ppm) Carbon NMR 128.8 128.4 40.0 -126.1 43.5- 138.8 120 50 40 140 130 110 100 90 80 70 60 Chemical Shift (ppm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (16 reviews)

Answered By

Antony Sang
I am a research and academic writer whose work is outstanding. I always have my customer's interests at heart. Time is an important factor in our day to day life so I am always time conscious. Plagiarism has never been my thing whatsoever. I give best Research Papers, Computer science and IT papers, Lab reports, Law, programming, Term papers, English and literature, History, Math, Accounting, Business Studies, Finance, Economics, Business Management, Chemistry, Biology, Physics, Anthropology, Sociology, Psychology, Nutrition, Creative Writing, Health Care, Nursing, and Articles.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the structure of the compound with molecular formula C 6 H 15 N that exhibits the following 1 H NMR and 13 C NMR spectra: 3 Proton NMR 3.0 2.8 2.6 2.4 2.2 2.0 1.8 1.6 1.4 1.2 1.0 0.8 0.6 0.4...
-
Propose a structure for a compound with molecular formula C 8 H 18 O that exhibits the following 1 H NMR and 13 C NMR spectra. Proton NMR 3.0 2.0 Chemical shift (ppm) 4.0 3.5 2.5 1.5 1.0 0.5 Carbon...
-
Compound A has molecular formula C 7 H 12 . Hydrogenation of compound A produces 2 methylhexane. Hydroboration-oxidation of compound A produces an aldehyde. Draw the structure of compound A, and draw...
-
You have recently been hired as a consultant for a personal financial planning firm. One of your first projects is creating a retirement plan for a couple, Tom and Helena Keeley. They have just...
-
Prove the identity. 1. nPn1 = nPn 2. nCn = nC0 3. nCn-1 = nC1 4. nCr = nPr / r! 5. Can your graphing utility evaluate 100P80? If not, explain why.
-
States have enormous flexibility in designing their Medicaid programs. States spend significant resources on optional services or mandatory services for optional populations. Even so, states complain...
-
Reproduce Figure 2.4 using geom_histogram(aes(y = ..density..)) as shown below for Example 2.5. ggplot_build() extracts the computed values for the histogram. Use the extracted vales to confirm that...
-
The balance sheet of Kishwaukee Corporation as of December 31, 2014, is as follows. Note 1: Buildings are stated at cost, except for one building that was recorded at appraised value. The excess of...
-
Robert is planning to open his own residential building design business. What nontax factors should Robert consider as he chooses his business entity? Discuss and explain whether the nontax factors...
-
Using two modulo-16 counters plus whatever other logic that is needed (including a gates and flip-flop), design a system with following counting sequence: 0 1 2 . 1523 45 .. 15 4 5 6 . 15 .. 12 13 14...
-
Using benzene as your only source of carbon atoms and ammonia as your only source of nitrogen atoms, propose a synthesis for the following compound: HN NH
-
Propose a mechanism for the following process: REN heat + N2 + CO2
-
For the following exercises, use a calculator to graph the function. Then, using the graph, give three points on the graph of the inverse with y-coordinates given. f(x) = x 3 x 2, y = 1, 2, 3
-
A parent and a subsidiary continue to exist as separate legal entities even though they are economically one reporting entity. Is this true or false?
-
Article-based Discussion Read the article Z Scores A Guide to Failure Prediction, by G.J. Eidleman, The CPA Journal, February 1995. Required: 1. What judgments are involved in the Z-score? 2. What...
-
To test whether a bonus plan will improve the monthly sale volume in units, the monthly sale volumes of six salespersons before and after a bonus plan were recorded. At 99 % confidence, determine...
-
A production manager is interested in the number of defects in batches derived from different production processes. He examines a random sample drawn from each process and records the following data:...
-
Refer to question 41 to find the power of a 10 % level test when the true population mean mileage is 36 miles per gallon. Question 41 An automobile manufacturer claims that a new car gets an average...
-
Consider the collision of a ball on an equal ball at rest. Compute the angle between the two final directions at non-relativistic speeds.
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Draw the structures of the following compounds. (a) 4-(1,1-dimethylethyl)octane (b) 5-(1,2,2-trimethylpropyl)nonane (c) 3,3-diethyl-4-(2,2-dimethylpropyl)octane
-
Without looking at the structures, give molecular formulas for the compounds in Problem 3-8 (a) and (b). Use the names of the groups to determine the number of carbon atoms, then use the (2n+2) rule.
-
Classify each hydrogen atom in the following compounds as primary (1), secondary (2), or tertiary (3). (a) Butane (b) Isobutene (c) 2-methylbutane (d) Cyclohexane (e) Norbornane (bicycle [2.2.1]...
-
Denise and Richard Deleon want to buy a house for $375,000. They can make a down payment of $100,000 from a combination of their RSPs ($25000 each under the Home Buyers program) and wedding gifts...
-
(a) Compare 'free cash flow to the firm' and 'free cash flow to equity' as inputs to discounted cash flow models. In your answer, you should (i) (ii) (iii) describe each model (including its inputs);...
-
In 1975 the price of a new house was $53,911. In 2020 the price of a new house is $180,916. How much has the price of housing increased on average per year over the time period in percentage terms?...

Study smarter with the SolutionInn App


