Use the bond energies in Table 17.1 and the accounting format shown in Section 17.5 to determine
Question:
Use the bond energies in Table 17.1 and the accounting format shown in Section 17.5 to determine whether these reactions are exothermic or endothermic:
H2 + Cl2 ¡ 2 HCl
2 HC‚CH + 5 O2 → 4 CO2 + 2 H2O
Table 17.1
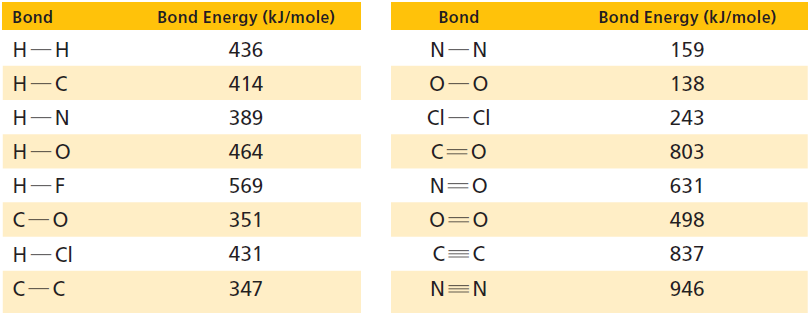
Transcribed Image Text:
Bond Energy (kJ/mole) Bond Energy (kJ/mole) Bond Bond Н-Н 436 159 Н-С 414 138 Cl-CI 389 Н—N 243 803 Н—О 464 C=0 Н-—F 569 N=0 631 351 498 Н— СІ 431 837 N=N 347 946
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a Energy to break bonds Energy released from bond formation H x H 43...View the full answer

Answered By

Amos Kiprotich
I am a wild researcher and I guarantee you a well written paper that is plagiarism free. I am a good time manager and hence you are assured that your paper will always be delivered a head of time. My services are cheap and the prices include a series of revisions, free referencing and formatting.
4.90+
15+ Reviews
21+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
Use the average bond energies in Table 4.3 to estimate ÎU for the reaction C 2 H 4 (g) + H 2 (g) C 2 H 6 (g). Also calculate ÎU o R from the tabulated values of ÎH o f for reactant...
-
The following table summarizes 204 endothermic reactions involving sodium bicarbonate. Let A denote the event that a reactions final temperature is 271 K or less. Let B denote the event that the heat...
-
2 Section 3 Date 4567 8 9 10 11 12 13 14 15 16 STRANK 21 22 23 Pharoah Inc. sells storage buildings of various sizes to homeowners and businesses. Respond to the requirements related to the following...
-
apple company: 1.what are key characteristics of the industry? 2. where is the company in its life cycle?
-
Arthur Wesson, an unmarried individual who is age 68, reports 2014 taxable income of $160,000. He records AMT positive adjustments of $40,000 and tax preferences of $35,000. a. What is Arthur's AMT?...
-
Solve the given systems of equations algebraically. 3x + 3y = 6 2 y 2 = 1
-
For each of the assets named in Problem 9.4 (ag), state both the MACRS-GDS property class, if applicable, and the specific depreciation method to be used. (e.g., 15-year Property; 150 percent DBSLH)...
-
Orange County Chrome Company manufactures three chrome-plated productsautomobile bumpers, valve covers, and wheels. These products are manufactured in two production departments (Stamping and...
-
(10m) 4. Explain the importance of LOFSA to the Malaysian economy and explain the steps to be taken by the LOFSA to revive the offshore markets in the endemic stage. (10m)
-
Brothers Harry and Herman Hausyerday began operations of their machine shop (H & H Tool, Inc.) on January 1, 2016. The annual reporting period ends December 31. The trial balance on January 1, 2018,...
-
Rank the following in order of increasing number of atoms: (a) 52 g of vanadium, V; (b) 52 g of chromium, Cr; (c) 52 g of manganese, Mn.
-
Bakers yeast contains a biological catalyst known as catalase, which catalyzes the transformation of hydrogen peroxide, H 2 O 2 , into oxygen, O 2 , and water, H 2 O. Write a balanced equation for...
-
The following credit sales are budgeted by Milford Company: May ....... $136,000 June ....... 200,000 July ....... 280,000 August ..... 240,000 The companys past experience indicates that 70% of the...
-
Today, Green Branch Coffee's employees began the process of forming a union. The Director of Human Resources wants you to discuss a few important facts about unions so that the coffee shop managers...
-
State and overall position regarding the meaning and function of leadership in organizations today.
-
To provide incentives, the NWC made the supervisors in Montreal and at the winter trading posts partners. A negative aspect of this organizational form was Group of answer choices the NWC was cash...
-
Although achieving horizontal and vertical fits are important for high-performance work systems, what do you consider more critical and why?
-
Think of the human resoruces organization that you are part of daily. Discuss how you communicate within this organization from a systems perspective. Describe the human resources organization and...
-
1. The yield in bushels from a grove of orange trees is given by Y = x(800 - x), where x is the number of orange trees per acre. How many trees will maximize the yield? 2. One of the early results in...
-
Answer the following two independent questions. a. MM Corporation is considering several proposed investments for the coming budget year. MM produces electrical apparatus for industrial complexes....
-
Liquid nitrogen at 77 K is stored in a cylindrical container having an inside diameter of 25 cm. The cylinder is made of stainless steel and has a wall thickness of 1.2 cm. Insulation is to be added...
-
The Fourier field equation in cylindrical coordinates is a. What form does this equation reduce to for the case of steady-state, radial heat transfer? b. Given the boundary conditions T = T i at r =...
-
Perform the same operations as in parts (a), (b), and (c) of Problem 16.1 with respect to a spherical system. Data From Problem 16.1 The Fourier field equation in cylindrical coordinates is a. What...
-
A liquid of density 1110 kg/m flows with speed 2.69 m/s into a pipe of diameter 0.28 m. The diameter of the pipe decreases to 0.05 m at its exit end. The exit end of the pipe is 8.48 m lower than the...
-
An investor can buy a tract of farmland today that he believes will sell for $50,000.00 in eight years. His required rate of return for this type of investment is 12% per annum compounded monthly....
-
If you started a simple business of washing windows, what do you feel would be your fixed costs? ( Please have current values for costs, when available )

Study smarter with the SolutionInn App


