Use the average bond energies in Table 4.3 to estimate ÎU for the reaction C 2 H
Question:
Table 4.3
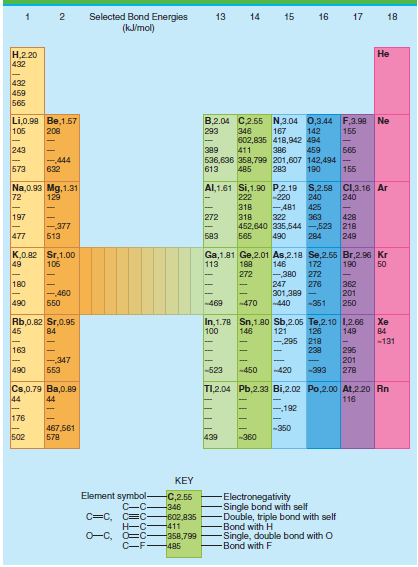
Transcribed Image Text:
Selected Bond Energies (klmol) 13 14 15 16 17 18 Н.220 432 Не 432 459 565 B,2.04 C2.55 N,3.04 0,3.44 F,3.98 Ne Li,0.90 Be,1.57 105 208 293 346 167 142 155 602,835 418,942 494 243 Б65 389 411 386 459 -444 536,636 358,799 201,607 142,494 155 573 632 613 485 283 190 Al,1.61 Si,1.90 P.2.19 S,2.58 C,3.16 Ar Na,0.93 Mg, 1.31 72 129 222 -220 240 240 -481 322 363 452,640 335,544 -523 218 318 425 197 272 318 428 -377 477 583 249 513 565 490 284 K,0.82 Sr,1.00 49 Ga,1.81 Ge,2.01 As,2.18 Se,2.55 Br,2.96 Kr 146 106 113 188 172 190 50 272 -380 272 180 247 362 201 250 276 301,389 - -440 -460 490 Б50 -469 470 -351 In,1.78 Sn,1.80 Sb,2.05 Te,2.10 1,2.66 121 -295 Rb,0.82 Sr,0.95 45 Xe 100 149 84 146 126 84 218 238 -131 163 296 201 278 -,347 553 490 -523 -420 450 -393 TI,2.04 Pb,2.33 Bi,2.02 Po,2.00o At,220 Rn Cs,0.79 Ba,0.89 44 116 44 192 176 -350 467,561 578 502 439 -360 KEY Element symbol- C-C C=C, C=C Electronegativity Single bond with self Double, triple bond with self Bond with H Single, double bond with O -Bond with F C,2.55 -346 602,835 -411 O-C, O=C- -358,799 C-F485
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
U R C C bond energy 6C H bond energy H H bond energy C C bond energy 4C H bond energy U R 346 kJ mo...View the full answer

Answered By

Nimlord Kingori
2023 is my 7th year in academic writing, I have grown to be that tutor who will help raise your grade and better your GPA. At a fraction of the cost on other sites, I will work on your assignment by taking it as mine. I give it all the attention it deserves and ensures you get the grade that I promise. I am well versed in business-related subjects, information technology, Nursing, history, poetry, and statistics. Some software's that I have access to are SPSS and NVIVO. I kindly encourage you to try me; I may be all that you have been seeking, thank you.
4.90+
360+ Reviews
1070+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Basing your answers on the bond dissociation energies in Table 4.3, calculate which of the following reactions are endothermic and which are exothermic: (a) (CH3)2CHOH + HF - (CH3)2CHF + H2O (b)...
-
Use the following data (in kJ/ mol) to estimate ÎH for the reaction S-(g) + e- S2-(g). Include an estimate of uncertainty. S(s) S(g) ÎH = 277 kJ/ mol S(g) + e- S-(g) ÎH = 200. kJ/...
-
The rates of many atmospheric reactions are accelerated by the absorption of light by one of the reactants. For example, consider the reaction between methane and chlorine to produce methyl chloride...
-
A jet is traveling westward with the sun directly overhead (the jet is on a line between the sun and the center of the Earth). How fast must the jet fly in order to keep the sun directly overhead?...
-
Which of the following statements is NOT CORRECT? (a) When a corporation's shares are owned by a few individuals who own most of the stock or are part of the firm's management, we say that the firm...
-
What are the three key features of cost accounting and cost management?
-
A data set of 50 comprehension scores (Comp Score). Complete the tasks and answer the questions below. a. Create a frequency distribution and a histogram for the set. b. Why did you select the class...
-
Some people like to pour beer gently down the side of the glass to preserve bubbles. Others, splash it down the center to release the bubbles into a foamy head and free the aromas. Champagne, however...
-
Concord Co. decides at the beginning of 2025 to adopt the FIFO method of inventory valuation. Concord had used the LIFO method for financial reporting since its inception on January 1, 2023, and had...
-
Giant pharmaceutical manufacturers such as Merck & Company and Pfizer have traditionally relied heavily on the doctor-to-patient channel to promote their products. In essence, this channel structure...
-
Using the protein DSC data in Problem P4.10, calculate the enthalpy change between the T = 288 K and T = 318 K. Give your answer in units of kJ per mole. Assume the molecular weight of the protein is...
-
In the compounds below, classify each bond as covalent, polar covalent, or ionic: a) NaBr b) NaOH c) NaOCH 3 d) CH 3 OH e) CH 2 O
-
The bookkeeper for Lowell's Country Music Bar went insane and left this incomplete balance sheet. Lowell's working capital is $90,000 and its debt to assets ratio is 40 percent. Required Complete the...
-
A loan requires a single payment of $4,000 at the end of three years. The loan's interest rate is 6%, compounded semiannually. How much was borrowed (rounded to the nearest dollar)? Group of answer...
-
If Acme Corporation issued $1,000,000 worth of 10-year, 9% coupon bonds payable on January 1 at $95 per $100 of face value, market interest rates were Select one: a. Less than 9% b. There is not...
-
Which of the following investments pays you the best return?Group of answer choicesAn investment paying interest of 11.6% compounded monthlyAn investment paying interest of 11.5% compounded...
-
How consumers and financial service organizations can benefit from using the Personal Property Securities Register, administered by the Australian Financial Security Authority.
-
What are the effective limits of production and profitability? Explain each briefly.
-
Find each sum or difference, if possible. -2 [2 4 6] +|-4 -6
-
What is the difference between adsorption and absorption?
-
Derive an expression for the rate of disappearance of a species A in a photochemical reaction for which the mechanism is: Hence, show that rate measurements will give only a combination of k2 and k3...
-
Photolysis of Cr (CO)6 in the presence of certain molecules M, can give rise to the following reaction sequence: Suppose that the absorbed light intensity is so weak that! «k4 [Cr (CO) 5M]....
-
Many enzyme-catalyzed reactions are consistent with a modified version of the Michaelis-Menten mechanism in which the second step is also reversible. (a) For this mechanism show that the rate of...
-
Ramirez Company installs a computerized manufacturing machine in its factory at the beginning of the year at a cost of $45,300. The machine's useful life is estimated at 10 years, or 403,000 units of...
-
14. (3 points) Write a program that ask the user for 1. their first name and 2. their last name, Enter first name: Matt Enter last name: Priem Hello Matt Priem! and then outputs a greeting similar to...
-
Given a sorted array 2, 5, 8, 12, 16, 18, 22, 25, 29, 32 and the following interpolation search algorithm. Show the steps of the algorithm when 25 is searched. Also, when 26 is searched. array a,...

Study smarter with the SolutionInn App


