Question: Find the range of K for stability for the unity feedback system of Figure P6.3 with K(s + 4)(s 4) (s +3) G(s) R(s) +
Find the range of K for stability for the unity feedback system of Figure P6.3 with
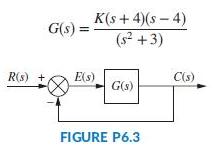
K(s + 4)(s 4) (s +3) G(s) R(s) + E(s) C(s) G(s) FIGURE P6.3
Step by Step Solution
3.44 Rating (154 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


