A 30-ft-high cylinder, cross-sectional area 1 ft 2 , has a massless piston at the bottom with
Question:
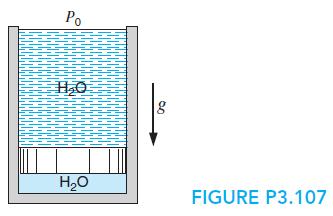
A 30-ft-high cylinder, cross-sectional area 1 ft2, has a massless piston at the bottom with water at 70 F on top of it, as shown in Fig. P3.107. Air at 540 R, volume 10 ft3 under the piston is heated so that the piston moves up, spilling the water out over the side. Find the total heat transfer to the air when all the water has been pushed out.
Transcribed Image Text:
Po H20 FIGURE P3.107
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
The total heat transfer to the air is 1068000 BTU The total heat tr...View the full answer

Answered By

Firoz K
I have extensive experience in education and tutoring, having worked as a tutor for the past three years in both group and individual settings. During my time as a tutor, I have successfully helped students improve their academic performance in a variety of subjects, including mathematics, science, language arts, and social studies. I have also developed and implemented personalized learning plans and differentiated instruction techniques to accommodate the individual needs of my students. Moreover, I have effectively communicated with parents and teachers to ensure that the students receive the best possible education and guidance. My strong organizational, communication, and problem-solving skills have enabled me to successfully collaborate with students, parents, and teachers in order to provide an effective and enjoyable learning experience.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A 0.500-kg slab of granite is heated so that its temperature increases by 7.40C. The amount of heat supplied to the granite is 2.93 kJ. Based on this information, what is the specific heat of granite?
-
As shown in fig 1.7, a manometer is attached to a tank of gas in which the pressure is 104.0 kPa. The manometer liquid is mercury, with a density of 13.59 g/cm3. If g = 9.81 m/s2 and the atmospheric...
-
As shown in Fig. 2, an air conditioner operating at steady state maintains a dwelling at 70 F on a day when the outside temperature is 90 F. If the rate of heat transfer into the dwelling through the...
-
ABC Client Preliminary Analytics - PBC Trial Balance December 31, 2019 Scope: Performance Materiality $210,000 and 10% 12/31/19 Balance 12/31/18 Account Class Balance Variance * Varlance Cash...
-
According to a report published by the Pew Research Center in February 2010, 61% of Millennial (Americans in their teens and 20s) think that their generation has a unique and distinctive identity (N...
-
Consider a version of the pipeline from Section 4.6 that does not handle data hazards (i.e., the programmer is responsible for addressing data hazards by inserting NOP instructions where necessary)....
-
Prove that, if \(\tau\) is an initial time with \(\mathbb{E}_{\mathbb{Q}}\left(1 / \alpha_{\infty}^{\tau} ight)
-
Value World produces canned chicken a la king. The chicken a la king passes through three departments: (1) Mixing, (2) Retort ( sterilization), and (3) Packing. In the Mixing Department, chicken and...
-
1. What specific marketing advantages does having pop-ups in Walmart provide for Sleep Country? How do you think these pop-ups will impact Sleep Country's iconic jingle "Why buy a mattress anywhere...
-
Jorge is a registered nurse at a facility that cares for about 80 elderly patients. Jorge has been at the facility longer than any of the other nurses and has his choice of schedule; he believes he...
-
A piston/cylinder arrangement with a linear spring similar to Fig. P3.105 contains R-134a at 60 F, x = 0.6 and a volume of 0.7 ft 3 . It is heated to 140 F, at which point the specific volume is...
-
Two rigid tanks are filled with water, as shown in Fig. P3.214. Tank A is 7 ft 3 at 1 atm, 280 F and tank B is 11 ft 3 at saturated vapor 40 psia. The tanks are connected by a pipe with a closed...
-
The surveys discussed in Exercise 27 also asked about weekly household expenditures for groceries. Here is a table with the results for the years 2008, 2009, and 2010: State and test an appropriate...
-
Ross filed a tax return for Allie. During the interview conducted by Ross, Allie stated that she had made a charitable contribution of artwork in the amount of $20,000 during the tax year, when in...
-
Juan purchased $100 of groceries using cash. After getting home, Juan notices that one of the items he purchased for $20 is rotten, so he returns it for a full refund. What journal entry would the...
-
Q: howtheUKcouldprotectthedomesticbikeindustryingeneraland not Brompton specically. Do we need to discuss Brompton?
-
I. Define the mapping L: F5 P3 (F) such that 24 =(-1+23+224 +25) + (2x1-25)t + (3x1 +3 +223 +55) where x1, x2, x3, 4, 5 EF. 1. Show that is a linear transformation (7 points). 2. Find a basis for...
-
How do you calculate student loan interest for someone that has $84601.55 in wages to place in the Schedule 1 line 21 Student loan interest deduction? Student loan interest was $1750.
-
A convex lens of power + 12 D is used as a magnifier to examine a wildflower. What is the angular magnification if the final image is at? (a) Infinity (b) The near point of 25 cm
-
2. In the circuit given in Figure 2, i,(t) = 5.67cos(5t)A and v (t) = 70.71 cos(5t 60) V a) Find the equivalent load impedance. State whether the load is inductive or capacitive. b) Calculate the...
-
One of the constitutional isomers of xylene was treated with sodium, methanol, and ammonia to yield a product that exhibited five signals in its 13 C NMR spectrum. Identify which constitutional...
-
Consider the equilibrium C 2 H 6 (g) C 2 H 4 (g) + H 2 (g). At 1000.K and a constant total pressure of 1.00 bar, C 2 H 6 (g) is introduced into a reaction vessel. The total pressure is held constant...
-
Consider the following two compounds. How would you distinguish between them using: a) IR spectroscopy? b) 1 H NMR spectroscopy? c) 13 C NMR spectroscopy?
-
Assigned a project from my CPA firm. Ihave to go into a new building and try to find as many pieces of furniture and fixtures from the building as I can.So for example, a door, a drop ceiling, a...
-
Retail Food Group shares suffered their biggest fall in nine years yesterday after UBS said changes to international accounting standards could cause a "material" impact on the company's debt...
-
The following transactions occurred during the twelve-month accounting period ending December 31, 2016 for Hammerhead, Inc. ("Corporation"). Be sure to include in your analysis any adjusting journal...

Study smarter with the SolutionInn App


