Describe a case in which the liquid and vapor curves in a diagram such as Figure 14-16
Question:
Describe a case in which the liquid and vapor curves in a diagram such as Figure 14-16 would converge into a single curve. Is such a case likely to exist?
Figure 14-16
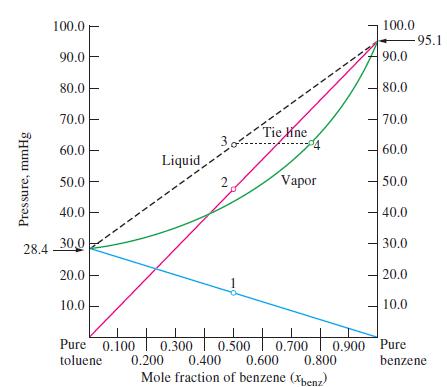
Transcribed Image Text:
Pressure, mmHg 100.0 90.0 80.0 70.0 60.0 50.0 40.0 28.4 30,0 20.0 10.0 Liquid Pure 0.100 toluene 2 1 Tie ne Vapor 100.0 90.0 80.0 70.0 60.0 50.0 40.0 30.0 20.0 -95.1 10.0 + to + 0.300 0.500 0.700 0.900 Pure 0.400 0.200 0.600 0.800 benzene Mole fraction of benzene (Xbenz)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
The liquid and vapor curves in a diagram such as Figure 1416 would converge into a single curve at t...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
We noted in Figure 14-17 that the liquid and vapor curves taken together outline a lens-shaped region when the normal boiling points of benzene-toluene solutions are plotted as a function of mole...
-
A constant-pressure R-134a vapor separation unit separates the liquid and vapor portions of a saturated mixture into two separate outlet streams. Determine the flow power needed to pass 6 L/s of...
-
A methanolwater feed stream is introduced to a vaporizer in which a molar fraction f of the feed is vaporized. The feed has a methanol mole fraction of x F = 0.4, and the vaporizer operates at a...
-
In the financial market, what causes a movement along the demand curve? What causes a shift in the demand curve?
-
The Current Designs staff has prepared the annual manufacturing budget for the rotomolded line based on an estimated annual production of 4,000 kayaks during 2013. Each kayak will require 54 pounds...
-
Force F acts perpendicular to the inclined plane. Determine the moment produced by F about point A. Express the result as a Cartesianvector. F = 400 N 4m,
-
One critical-thinking skill is a heightened awareness of the danger of reaching a conclusion prior to acquiring missing information that were it known would have a reasonable probability of altering...
-
Polk and Stoneman is a public accounting firm that offers two primary services, auditing and tax return preparation. A controversy has developed between the partners of the two service lines as to...
-
A certain low-loss non-magnetic dielectric material has a relative permittivity (er) of 2.5 and a loss tangent of 0.004. What is the phase constant b of a plane wave of frequency 11.3 GHz?
-
Two of the substances listed here are highly soluble in water, two are only slightly soluble in water, and two are insoluble in water. Indicate the situation you expect for each one. (a) iodoform,...
-
A solution is prepared by dissolving 95 g NH 4 Cl in 200.0 g H 2 O at 60 C. (a) What mass of NH 4 Cl will recrystallize when the solution is cooled to 20 C? (b) How might we improve the yield of NH 4...
-
Annies Barbecue Restaurant prepares two primary food items fresh each morning, pulled pork barbecue and beef brisket barbecue, which are sold as a part of different menu items, including sandwiches...
-
Your daughter is currently twelve years old. You anticipate that she will be going to college in six years. You would like to have $127,000 in a savings account to fund her education at that time. If...
-
Define the communication skills applicable to listening and feedback.
-
You are thinking of retiring. Your retirement plan will pay you either $100,000 immediately on retirement or $140,000 five years after the date of your retirement. Which alternative should you choose...
-
Explain the terms criterion and predictor with respect to the validity of selection methods.
-
What form does communication as transactions take?
-
The following unadjusted trial balance is for Power Demolition Company as of the end of its April 30, 2013, fiscal year. The April 30, 2012, credit balance of the owners capital account was $46,900,...
-
When a company has a contract involving multiple performance obligations, how must the company recognize revenue?
-
Financing with convertible debt is especially appropriate for small, rapidly growing, or risky companies. Explain why.
-
The Pork Barrel Company has issued three-year warrants to buy 12 percent perpetual debentures at a price of 120 percent. The current interest rate is 12 percent and the standard deviation of returns...
-
The B.J. Services warrant is described in Section 23.1. How would you use the BlackScholes formula to compute the value of the warrant immediately after its issue, assuming a stock price of $19 and a...
-
Fix some a > 0. Consider the function G : (0,) (0,) given by a G(y) == y + (a) Show that G is continuously differentiable, and that G' (y) < 0 for y < a and G' (y) > 0 for y> a. Use this to show that...
-
Brandeis, Inc has a 45-day accounts payable period. The firm has expected quarterly sales of $2,400, $2,800, $3,600, and $4,200, respectively, for the next calendar year. The cost of goods sold for a...
-
The 100 kg plate is held in position by cables AB, AC and AD as shown. A vertical force P equal to the weight of the plate is applied at ring A to suspend it. Determine the tension in the 3 cables...

Study smarter with the SolutionInn App


