For each of the following molecules, determine the hybridization of each carbon atom, the total number of
Question:
For each of the following molecules, determine the hybridization of each carbon atom, the total number of carbon–carbon bonds and the total number of carbon–carbon π bonds. Also, among the sp3-hybridized carbon atoms, which ones are primary (1°), secondary (2°), tertiary (3°), or quaternary (4°)?
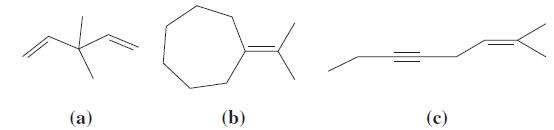
Transcribed Image Text:
(a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Molecule 1 Hybridization of each carbon atom C1sp3 C2sp2 C3sp2 Total number of carboncarbon bonds4 Total number of carboncarbon bonds2 Among the sp3hybridized carbon atomsC1 is primary Molecule 2 Hybridization of each carbon atom C1sp3 C2sp3 C3sp3 C4sp2 Total number of carboncarbon bonds7 Total number of carboncarbon bonds2 Among the sp3hybridized carbon atomsC1C2and C3 are primaryand C4 is secondary Molecule 3 Hybridization of each carbon atom C1sp3 C2sp3 C3sp3 C4sp2 Total number of carboncarbon bonds8 Total number of carboncarbon bonds3 Among the sp3hybridized carbon atomsC1C2and C3 are primaryand C4 is secondary Summary Molecule Hybridization of each carbon atom Total number of carboncarbon bonds ...View the full answer

Answered By

John Aketch
I am a dedicated person with high degree of professionalism, particularly in academic writing. My desire is to is to make students excel in their academic endeavor.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
1. Prepare income statements for both Garcon Company and Pepper Company. 2. Prepare the current assets section of the balance sheet for each company.
-
In Exercises 133136, determine whether each statement makes sense or does not make sense, and explain your reasoning. and one piece of my graph is a single point. I graphed f(x) = 2 if if 3 if x 4 x...
-
The equations in Exercises 6980 combine the types of equations we have discussed in this section. Solve each equation. Then state whether the equation is an identity, a conditional equation, or an...
-
In terms of Federal tax consequences, why are lifetime transfers to charity preferable to testamentary transfers?
-
Wal-Mart is the worlds retailer, employing more than 2 million associates worldwide, including more than 1.4 million in the United States. Wal-Mart has more than 7,870 stores, including about 890...
-
Under the current rules of both U.S. GAAP and IFRS, companies must charge research expenditures directly to expense. Some accountants believe that companies should be allowed to recognize some such...
-
Its Intoxicating Inc., is a Pennsylvania corporation that manufactures and distributes cosmetic products to various retailers. Maritim is a German company that owns and operates hotels throughout...
-
The following is Sullivan Corp.'s comparative balance sheet accounts at December 31, 2010 and 2009, with a column showing the increase (decrease) from 2009 to 2010. Additional information: 1. On...
-
Following are the results from two different simple regression analyses, estimating the costs of the purchasing department using number of purchase orders and number of vendors as potential cost...
-
Determine the configuration, R or S, of each chiral carbon atom in the molecules that follow. (Ph represents a phenyl group.) () HO. H H CI CH3 CH3 Ci M H (b) HO Ph - H Ph
-
Ephedrine (shown below) is used as a decongestant in cold remedies. (a) Name the functional groups of ephedrine. (b) What is the hybridization of the numbered carbon atoms and the nitrogen atom? (c)...
-
The authors of an International Monetary Fund working paper state that consumers lose more from a tariff than producers gain, so there is a deadweight loss. Briefly explain what the authors mean by...
-
O Macmillan Learning Solid ammonium chloride, NH Cl, is formed by the reaction of gaseous ammonia, NH3, and hydrogen chloride, HCl. NH3 (g) + HCl(g) NHCl(s) A 6.00 g sample of NH3 gas and a 6.00 g...
-
The Rydberg equation gives us the mathematical relationship between: A) an explanation for the continuous range of energy values associated with atomic spectra B) wavelength and frequency C) electron...
-
18. If 5.0 moles of calcium reacts with excess oxygen as shown, 2 Ca(s) + O2(g) 2CaO(s) AHxn= -1,270 kJ a. The reaction releases 6,350 kJ. b. The reaction absorbs 6,350 kJ. c. The reaction releases...
-
When aluminum is placed in concentrated hydrochloric acid, hydrogen gas is produced. 2 Al(s) + 6 HCl(aq) ->> 2 AICI, (aq) + 3 H(g) What volume of H(g) is produced when 7.20 g Al(s) reacts at STP?...
-
Draw a roadmap and write a plan to find the mass ( g ) of the products based on the given amount of reactants. 2 H 2 O 2 - > 2 H 2 O + O 2 given: H 2 O 2 = 4 9 . 4 mg
-
We point out in this chapter that the organizational form of a business will determine the amount of regulation that business experiences. Furthermore, business owners will have different rights and...
-
An item of depreciable machinery was acquired on 1 July 2009 for $120,000 by cash It is expected to have a useful life of 10 years and zero salvage value On 1 July 2012, it was decided to revalue the...
-
What happens if a company has a constant g that exceeds its rs? Will many stocks have expected g > rs in the short run (i.e., for the next few years)? In the long run (i.e., forever)?
-
Assume that Temp Force has a beta coefficient of 1.2, that the risk-free rate (the yield on T-bonds) is 7%, and that the market risk premium is 5%. What is the required rate of return on the firms...
-
Assume that Temp Force is a constant growth company whose last dividend (D0, which was paid yesterday) was $2.00 and whose dividend is expected to grow indefinitely at a 6%rate. What is the firms...
-
To analyze the risk, or volatility, associated with investing in a certain common stock, consider a sample of the eight quarterly percent total returns. The percent total return includes the stock...
-
Accounting for pension and post-employment benefits is a complex area of financial reporting, requiring companies to accurately measure and disclose their obligations to current and former employees....
-
What are the sex-specific differences in hormonal regulation of reproductive function, metabolism, and behavior, and how do these differences contribute to sexual dimorphism in health and disease?

Study smarter with the SolutionInn App


