For the reaction 2 NO 2 (g) 2 NO(g) + O 2 (g), K c =
Question:
For the reaction 2 NO2(g) ⇌ 2 NO(g) + O2(g), Kc = 1.8 x 10-6 at 184 °C. What is the value of Kp for this reaction at 184 °C, for pressures expressed in atmospheres?
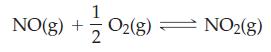
Transcribed Image Text:
(8)702 + NO(g) + O2(g) NO2(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Python import math Given Kc and the balanced chemical equation calculate Kp ...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The reaction 2 NO2 2 NO + O2 has the rate constant. k = 0.63 M-1s-1. Based on the units for k, is the reaction first or second order in NO2? If the initial concentration of NO2 is 0.100M, how would...
-
Hugo read a blog about brainstorming and decided that this would be a great way for his customer relations team at Royal Caribbean Cruise lines to generate new ideas for assuring passengers that the...
-
What are two cash management issues for a clothing business? explain Cash Management Issues Working capital management Need for accurate cash flow forecasting Evaluate capital expenditure Optimize...
-
Read each case carefully and, as determined in the Internal Revenue Code of Puerto Rico, identify the deductions, calculate the amount of deductions and what the determined contribution would be. and...
-
Litho-Print is considering two possible capital structures, A and B, shown in the following table. Assume a 40% tax rate. a. Calculate two EBIT-EPS coordinates for each of the structures by selecting...
-
Marcia Samms claimed that David Eccles had repeatedly and persistently called her at various hours, including late at night, from May to December, soliciting her to have illicit sexual relations with...
-
Oncology Tech is a manufacturer of cancer therapy devices called compensators. CMS was a company that sold cancer treatment planning software for use in purchasing cancer treatment devices. In 2006,...
-
Churchill Co. uses a job order cost system. The following data summarize the operations related to production for November: a. Materials purchased on account, $ 528,000. b. Materials requisitioned, $...
-
A bug sits on a record moving at a constant angular speed of 0 = 3.5 rad/s. Starting at t = 0 s, the record player is turned off and is given a constant angular acceleration of = 1.0 rad/s 2 . The...
-
Continuous removal of one of the products of a chemical reaction has the effect of causing the reaction to go to completion. Explain this fact in terms of Le Chteliers principle.
-
Exactly 1.00 mol each of CO and Cl 2 are introduced into an evacuated 1.75 L flask, and the following equilibrium is established at 668 K. For this equilibrium, calculate (a) The partial pressure of...
-
Joints are places where bones meet. Many of them, such as the ball-and-socket joints in your shoulders and hips, are bathed with synovial fluid, a viscous substance resembling the white of an egg....
-
Sam purchased a 10% capital and profit interest in the partnership in 2021 he does not participate in the partnership's business Sam has no passive income in the current year it's distributive share...
-
How do environmental factors, such as urbanization, environmental degradation, and climate change, interact with social structures and systems, exacerbating or mitigating issues of inequality, health...
-
The nominal accounts of Mount Dora Nursing Home Inc. for the year ended December 2022 are listed below: Depreciation Insurance expense Interest expense Medical supplies and drugs Other revenue Net...
-
A recent survey resulted in 48 students stating that Economic Statistics is Easy, 18 students stated that Economic Statistics was Hard and the remaining 54 students stated that Economic Statistics...
-
What are the prevailing cultural trends pertaining to the elderly population in contemporary society?
-
Buzz can be created in a variety of ways, for example, advertising. Explain how advertising can be used to generate buzz. What are some of the dangers of this method?
-
A Firm intends to invest some capital for a period of 15 years; the Firm's Management considers three Options, each consisting of purchasing a machinery of a specific brand, different for each...
-
Pet Joy Wholesale Inc., a pet wholesale supplier, was organized on May 1, 2010. Projected sales for each of the first three months of operations are as follows: May $360,000 June 450,000 July 600,000...
-
Office Mate Supplies Inc. has cash and carry customers and credit customers. Office Mate estimates that 25% of monthly sales are to cash customers, while the remaining sales are to credit customers....
-
Excel Learning Systems Inc. was organized on May 31, 2010. Projected selling and administrative expenses for each of the first three months of operations are as follows: June $117,400 July 110,500...
-
Suppose you want to have $400,000 for retirement in 20 years. Your account earns 8% interest. a) How much would you need to deposit in the account each month? $ b) How much interest will you earn? $
-
9. Given the class data, complete parts a-e with a class width of 20 where appropriate (5 points each). a. frequency distribution b. frequency polygon c. histogram d. stem and leaf plot e. What is...
-
Prepare journal entries to record the following transactions. a. Purchased $490 of supplies on credit. b. Completed $690 of work for a client on credit. c. Paid $490 cash towards the amount owed from...

Study smarter with the SolutionInn App


