From Figure 12-18, estimate (a) The vapor pressure of C 6 H 5 NH 2 at 100C
Question:
From Figure 12-18, estimate
(a) The vapor pressure of C6H5NH2 at 100°C
(b) The normal boiling point of C6H5CH3.
Figure 12-18
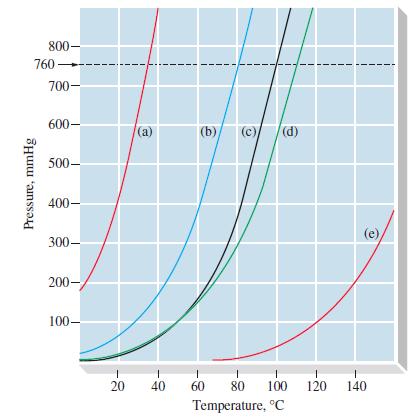
Transcribed Image Text:
800- 760- Pressure, mmHg 700- 600- 500- 400- 300- 200- 100- 20 (a) (b) (c) (d) 1 1 1 40 60 80 100 Temperature, °C 1 120 1 140
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a It appears that the vapor pressure of C6H5NH2 ...View the full answer

Answered By

Collins Omondi
I have been an academic and content writer for at least 6 years, working on different academic fields including accounting, political science, technology, law, and nursing in addition to those earlier listed under my education background.
I have a Bachelor’s degree in Commerce (Accounting option), and vast knowledge in various academic fields Finance, Economics, Marketing, Management, Social Science, Women and Gender, Business law, and Statistics among others.
4.80+
4+ Reviews
16+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Procurement of commodities is a critical activity for a company's survival, particularly in the manufacturing industry. It is essential in the industry because it standardizes the procurement of...
-
The normal boiling point for acetone is 56.5oC. At an elevation of 5300 ft, the atmospheric pressure is 630 torr. What would be the boiling point of acetone (Hvap = 32.0 kJ/mol) at this elevation?...
-
The vapor pressure of liquid benzene is 20,170 Pa at 298.15 K, and Î H vaporization =30.72 kJ mol -1 at 1 atm pressure. Calculate the normal and standard boiling points. Does your result for...
-
Goddard Company has used the FIFO method of inventory valuation since it began operations in 2015. Goddard decided to change to the average cost method for determining inventory costs at the...
-
Fred Lane, who sells boats, motors, and trailers, sold a boat, motor, and trailer to John Willis in exchange for a check for $6,285.00. The check was not honored when Lane attempted to use the funds....
-
What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities. (b) CH |(a) (c) H CHCH2CH2H2H2 C CHCH-CH
-
What is the expectations gap?
-
Designing a Balanced Scorecard strategy map for an auto parts manufacturing company Domestic Auto Parts (DAP), 16 a $1 billion subsidiary of a U.S. auto parts manufacturing company, manufactured and...
-
The eigenvalues, eigenvectors in Question-1 needs to be calculated by hand (not by computer). Questions-1: (9 mark) A car rental company has three locations. Every customer can rent from and return...
-
Use data in Figure 12-20 to estimate (a) The normal boiling point of aniline; (b) The vapor pressure of diethyl ether at 25 C. Figure 12-20 P 6.75 - 6.50- 6.25 6.00- 5.75- 5.50- 5.25 5.00 4.75 - 4.50...
-
A 50.0 g piece of iron at 152 C is dropped into 20.0 g H 2 O(l) at 89 C in an open, thermally insulatedcontainer. How much water would you expect to vaporize, assuming no water splashes out? The...
-
In Problems 530, a. Classify the sequences as arithmetic, geometric, Fibonacci, or none of these. b. If arithmetic, give d; if geometric, give r; if Fibonacci, give the first two terms; and if none...
-
3. (3 pts) Rearrange the recursive algorithm on linked list to non-recursive one. Function func (n) Input: A node n Output: A node else if n is null then return head tfunc (n.next) n.next null t.next...
-
Goodman, Pinkman, and White formed a partnership on January 1, 2024, and made capital contributions of $125,000 (Goodman), $175,000 (Pinkman), and $250,000 (White), respectively. With respect to the...
-
Provide two quantities data sources (metrics) that would provide information that would assist Netflix in making decisions. explain what discoveries could be made based on this data and why it would...
-
From the items below, explain three (3) management and communication techniques you can use to lead, mentor and motivate teams in the property industry. Actively listening Maintain silence Using...
-
What perspective on leadership focuses on the role communication plays in influencing and directing groups?
-
Manufacturers Southern leased high-tech electronic equipment from Edison Leasing on January 1, 2013. Edison purchased the equipment from International Machines at a cost of $112,080. Related...
-
Reduction in sales All of the above 29. Belt of an electric motor is broken, it needs a. Corrective maintenance b. Scheduled maintenance c. Preventive maintenance d. Timely maintenance. 30. The...
-
Capital Gains versus Income Consider four different stocks, all of which have a required return of 18 percent and a most recent dividend of $4.50 per share. Stocks W, X, and Y are expected to...
-
Stock Valuation most corporations pay quarterly dividends on their common stock rather than annual dividends. Barring any unusual circumstances during the year, the board raises, lowers, or maintains...
-
Non constant Growth Storico Co. just paid a dividend of $2.75 per share. The company will increase its dividend by 20 percent next year and will then reduce its dividen4 growth rate by 5 percentage...
-
Manufacturing costs for Blossom Company for selected months are as follows. (Assume all raw materials used were direct materials.) Indicate the missing amounts. April Beginning work in process...
-
The Shirt Shop had the following transactions for T-shirts for Year 1, its first year of operations: Jan. 20 Purchased Apr. 21 Purchased July 25 330 units @ $ 4 = 80 units @ $ 6 = $1,320 480...
-
Engberg Company installs lawn sod in home yards. The company's most recent monthly contribution format income statement follows: Sales Variable expenses Contribution margin Fixed expenses Net...

Study smarter with the SolutionInn App


