How would the Balmer equation (8.4) have to be modified to predict lines in the infrared spectrum
Question:
How would the Balmer equation (8.4) have to be modified to predict lines in the infrared spectrum of hydrogen?
Eq. 8.4
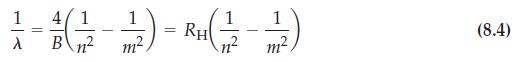
Transcribed Image Text:
1 4/1 1 - = - 3 (→ ) - ²+(²² - 2) RH A B 2 m² nº m (8.4)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
ANSWER The Balmer equation you provided denoted as Equation 84 describes the wavelengths of the spec...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
345+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
LIST-I contains compounds and LIST-II contains reactions. LIST-I (1) HO (II) (III) (IV) Mg(OH)2 BaCl CaCO3 (P) (Q) (R) LIST-II Mg(HCO3) + Ca(OH) BaO,+H,SO4 Ca(OH) + MgCl BaO, +HCI (T) Ca(HCO3)2 +...
-
In 2006 - only two years after the launch of Facebook, and one year after the launch of YouTube - Time magazine named "You" its Person of the Year. In the new information age, the cover declared,...
-
(a) How would Fig. 1.9 have to be modified to show boundary surfaces for the 2s and the 3p wavefunctions of a one-electron species? (b) The probability of finding the electron of a ground-state...
-
For the manipulator shown, find the twist coordinates f; at zero con- figuration and 9st (0) using the product of exponentials approach. Use the base and tool frames shown. (b) (10 points) Find efi0i...
-
Is it the right time to enter the Indian retail market? If so, what is the best entry mode?
-
The free-vibration response of a spring-mass system is observed to have a frequency of 2 \(\mathrm{rad} / \mathrm{s}\), an amplitude of \(10 \mathrm{~mm}\), and a phase shift of \(1 \mathrm{rad}\)...
-
A bridge is found to vibrate violently when a vehicle, producing a harmonic load of magnitude \(600 \mathrm{~N}\), crosses it. By modeling the bridge as an undamped spring-mass system with a mass...
-
Springtime Paints makes quality paint in one production department. Production begins with the blending of various chemicals, which are added at the beginning of the process, and ends with the...
-
1. A curling stone sliding on ice undergoes a constant acceleration of 5.1 cm/s [E] as it travels from initial position before coming to rest. When the stone was released, it had an initial velocity...
-
You have just been hired as a brand manager at Kelsey-White, an American multinational consumer goods company. Recently the firm invested in the development of K-W Vision, a series of systems and...
-
What is E for the transition of an electron from n = 6 to n = 3 in a hydrogen atom? What is the frequency of the spectral line produced?
-
Use the Balmer equation (8.4) to determine (a) The frequency, in s -1 , of the radiation corresponding to n = 5; (b) The wavelength, in nanometers, of the line in the Balmer series corresponding to n...
-
Baxter, Inc., has 25,000 shares of $1.35 preferred stock outstanding in addition to its common stock. The $1.35 designation means that the preferred stockholders receive an annual cash dividend of...
-
RCF to Debt = retained cash flow / debt Please used Bunge's financial statements from years 2017 to 2021. find financial statement for Bunge here...
-
5. Do some internet research to determine what happened to Genzyme in the years following the case (from May 2009) and where it is now. Be sure to cite your sources. I
-
OPERATING Income Margin = EBT / Sales Please do these calculations based on the income statement from years 2017-2022 please find financial statement for Bunge here...
-
Food sales are $82,800 and Beverage Sales are $36,900. If food cost is 32% and Beverage cost is 25%, Calculate Gross Profit.
-
5. You want to speculate in the forward market by either buying or selling 1,000,000. Suppose that the one-year forward rate is $1.10/, and the current spot rate is $1.30/. You expect the future spot...
-
Branded Products, Inc., based in Oakland, California, is a leading producer and marketer of household laundry detergent and bleach products. About a year ago, Branded Products rolled out its new...
-
Audrey purchases a riding lawnmower using a 2-year, no-interest deferred payment plan at Lawn Depot for x dollars. There was a down payment of d dollars and a monthly payment of m dollars. Express...
-
Describe the five-step decision-making process?
-
Distinguish planning decision from control decision?
-
What three guidelines help management accountants provide the most value to managers?
-
Write a program to display the following 1,5,13,25,41 The number needs to be generated automatically.
-
Perform the following arithmetic operations in their given bases. Explain or show the carries or borrows used whenever necessary. Also specify if you are switching the operands whenever necessary....
-
For any string w, let contract(w) denote the string obtained by collapsing each maximal substring of equal symbols to one symbol. For example: contract(010101) = 010101 contract(001110) = 010...

Study smarter with the SolutionInn App


