Lead metal is added to 0.100 M Cr 3+ (aq). What are [Pb 2+ ], [Cr 2+
Question:
Lead metal is added to 0.100 M Cr3+(aq). What are [Pb2+], [Cr2+], and [Cr3+] when equilibrium is established in the reaction?
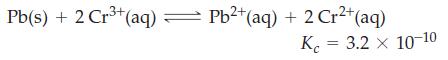
Transcribed Image Text:
3+ Pb(s) + 2 Cr³+ (aq) = Pb²+ (aq) + 2 Cr²+ (aq) K 3.2 x 10-10 =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The reaction is as follows Pbs 2 Cr3aq Pb2aq 2 Cr2aq The equilibrium constant is Kc 32 ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
When magnesium metal is added to a beaker of HCl(aq), a gas is produced. Knowing that magnesium is oxidized and that hydrogen is reduced, write the balanced equation for the reaction. How many...
-
An excess of zinc metal is added to 50.0 mL of a 0.100 M AgNO3 solution in a constant-pressure calorimeter like the one pictured in Figure 6.9. As a result of the reaction The temperature rises from...
-
Let us explore a reaction with a limiting reactant. Here, zinc metal is added to a flask containing aqueous HCl, and H 2 gas is a product. The three flasks each contain 0.100 mol of HCl. Zinc is...
-
Herbs Pty Ltd is considering investing in a new herb packaging machine. The machine is estimated to cost $80,000 which can last for 7 years before it becomes too costly to maintain and can be sold...
-
Wells and Associates has EBIT of $67,500. Interest costs are $22,500, and the firm has 15,000 shares of common stock outstanding. Assume a 40% tax rate. a. Use the degree of financial leverage (DFL)...
-
Scott, manufacturer of a carbonated beverage, entered into a contract with Otis, owner of a baseball park, whereby Otis rented to Scott a large signboard on top of the center field wall. The contract...
-
Defendant Monty J. Person began working for Garage Solutions, LLC, in March 2015. Three months into his employment, Person was sent by the owner of Garage Solutions, Mark Fontenot, to Rexburg, Idaho,...
-
After consulting with Butch Radner, George Monohan was able to determine the activity times for constructing the weed-harvesting machine to be used on narrow rivers. George would like to determine...
-
what are three techniques for processing BI DATA? explain clearly REPORTING ANALYSIS, RFM ANALYSIS,ONLINE ANALYTICAL PROCESSING,DATA MINING ANALYSIS?
-
One sketch below represents an initial nonequilibrium mixture in the reversible reaction Which of the other three sketches best represents an equilibrium mixture? Explain. Kc = 4.0 (8) (8) + (8)
-
One sketch below represents an initial nonequilibrium mixture in the reversible reaction Which of the other three sketches best represents an equilibrium mixture? Explain. 2 NO(g) + Br(g) = 2 NOBr(g)...
-
Locate the description of the ASTM Standard A992 structural steel that is commonly used for rolled-steel beam shapes. Determine how to acquire a copy of the standard.
-
Garvin Enterprises bonds currently sell for $1,100. They have a 6-year maturity, an annual coupon of $95, and a par value of $1,000. What is their current yield? Enter your answer rounded to two...
-
A 72-kg astronaut floating in space in a M = 118 -kg MMU (manned maneuvering unit) experiences an acceleration of 3.00102 m/s2 when he fires one of the MMU's thrusters. Part A. If the speed of the...
-
Given that P(A)=1, P(B)=, and P(A and B)=12, find P(A or B).
-
Your company has three million shares of common stock outstanding with a current market price of $28. RE = 14.5%. There are also 50,000 bonds outstanding with an 8 percent annual coupon, 18 years to...
-
The outcome of a money market hedge for C$200,000 6-month accounts receivable is when the spot rate is $0.9925 and the U.S and C$ interest rates are 3.00% and 3.5% respectively?
-
Three items in parallel each having a reliability of 0.90 have an overall system reliability of 0.729. True ______ False______
-
A. Select a recent issue (paper or online) of Report on Business Magazine, Canadian Business Magazine (online only), Bloomberg Businessweek, Fast Company, The Economist, or another business magazine....
-
McLean Company produces a product that requires three standard gallons per unit. The standard price is $18.50 per gallon. If 2,500 units required 8,000 gallons, which were purchased at $18.00 per...
-
Norris Company produces a product that requires 3.5 standard hours per unit at a standard hourly rate of $12 per hour. If 500 units required 1,500 hours at an hourly rate of $11.50 per hour, what is...
-
McLean Company produces a product that requires two standard hours per unit at a standard hourly rate of $18 per hour. If 2,500 units required 5,500 hours at an hourly rate of $19 per hour, what is...
-
At the beginning of the year, Yancey made the following estimates: Direct labor-dollars to support all productions Fixed overhead cost Variable overhead cost per direct labor-dollar Required: 1....
-
I specifically need help with what caused malware.hidden to be created and what caused the batch files in the recycle bin and the example videos folder in the Public users files to be created. I am...
-
What are the major differences between a list and a set in Java? How can you determine when to implement a list versus a set for an application? Provide an example code segment in Java that...

Study smarter with the SolutionInn App


