Refer only to the periodic table on the inside front cover, and arrange the following species in
Question:
Refer only to the periodic table on the inside front cover, and arrange the following species in order of increasing size: K+, Cl-, S2-, and Ca2+.
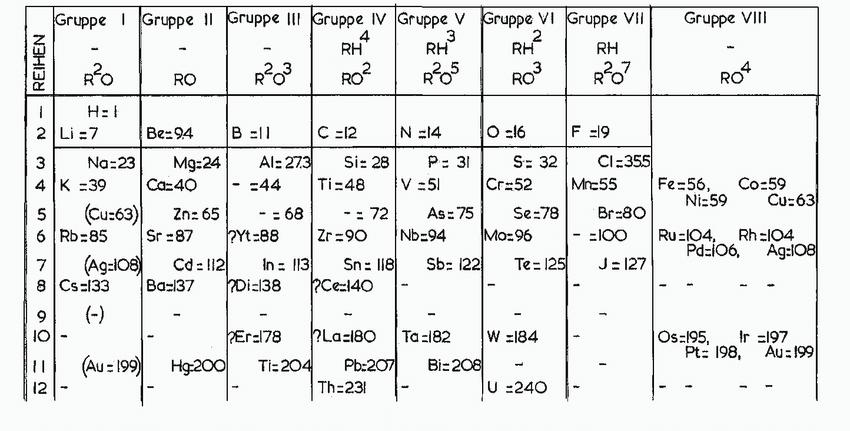
Transcribed Image Text:
REIHEN 3 A W 1 2 Li=7 4 K 56 94 Gruppe 1 Gruppe 11 Gruppe III Gruppe IV Gruppe V Gruppe VI Gruppe VII RH²³ 3 2.5 R²3 7 9 10 12 M 6 Rb 85 R²0 - H=1 - 8 Cs-133 (-) (Au=199) (Cu=63) Na:23 =39 Ca=40 (Ag=108) - RO Be=94 Mg:24 Cd: 112 B =11 Ba=137 - Al: 273 Zn: 65 Sr=87 ?Yt=88 =44 -=68 RH RO² Di=138 C =12 Si: 28 Ti=48 N=14 P= 31 V = 51 Hg:200 Ti=204 Pb:207 Th=231 -= 72 Zr=90 Nb=94 As: 75 In = 113 Sn: 118 Sb: 122 ?Ce=140 RH² 2 RO³ Bi-208 O=16 S= 32 Cr=52 Se=78 Mo=96 Er=178 ?La-180 Ta=182 W =184 Te = 125 L U =240 RH R²O7 F =19 CI=355 Mr-55 Br=80 - =100 J=127 Gruppe VIII Fe=56, RO4 Ni:59 Co=59 Cu:63 Ru-104, Rh=104 Pd=106, Ag:108 Os=195, Ir =197 Pt: 198, Au 199
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Analyze The key lies in recognizing that the four species are isoelectron...View the full answer

Answered By

Abigael martinez
I have been a tutor for over 3 years and have had the opportunity to work with students of all ages and backgrounds. I have a strong belief that all students have the ability to learn and succeed if given the right tools and support. I am patient and adaptable, and I take the time to get to know each student's individual learning style in order to best support their needs. I am confident in my ability to help students improve their grades and reach their academic goals.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) Refer only to the periodic table on the inside front cover, and arrange the following species in order of increasing size: Ti 2+ , V 3+ , Ca 2+ , Br - , and Sr 2+ . (B) Refer only to the periodic...
-
Refer only to the periodic table on the inside front cover, and arrange the following ionization energies in order of increasing value: the first ionization energy of F; the second ionization energy...
-
(A) Refer to the periodic table on the inside front cover, and arrange the following in the expected order of increasing first ionization energy Cl, K, Mg, S. (B) Refer to the periodic table on the...
-
The wheelbarrow and its contents have a center of mass at G. If F = 100 N and the resultant moment produced by force F and the weight about the axle at A is zero, determine the mass of the...
-
a. Calculate the approximate annual rate of return on investment of the following cash discount terms: 1. 1/15, n30. 2. 2/10, n60. 3. 1/10, n90. b. Which of these terms, if any, is not likely to be a...
-
Charlie Stork rented a truck for his business on two previous occasions. Since he will soon be renting a truck again, he would like to analyze his bills and determine how the rental fee is...
-
Refer to Samsungs financial statements in Appendix A. Compute its debt ratio as of December 31, 2015, and December 31, 2014. Data From Samsung Financial Statement Appendix A Samsung Electronics Co.,...
-
Meena Chavan Corp.s computer chip production process yields DRAM chips with an average life of 1,800 hours and = 100 hours. The tolerance upper and lower specification limits are 2,400 hours and...
-
An aluminum-alloy rod has a length of 9.2293 cm at 20.00C and a length of 9.2767 cm at the boiling point of water. (a) What is the length of the rod at the freezing point of water? (b) What is the...
-
(A) Use the periodic table on the inside front cover to predict which is the smallest atom: As, I, or S. (B) Which of the following atoms do you think is closest in size to the Na atom: Br, Ca, K, or...
-
Unpaired electrons are found in only one of the following species. Indicate which one, and explain why: F - , Ca 2+ , Fe 2+ , S 2- .
-
Find, if possible, AB and BA. If it is not possible, explain why. A = || -2 17 0 3 -1 02 2-1 B = [4 -15]
-
(a) The complex shown below is the first example of a Pd(IV) complex containing a nitrosyl ligand (see also structure 20.9 for another view of the tridentate ligand). On the basis of the assignment...
-
Stare at the image below and attempt to flip the stack of blocks upside down. If you cannot see a second perception, stare at the point where the three blocks come together and mentally pull the...
-
Using perturbation theory, derive the following expression for the matrix element pertinent to radiationless relaxation. How does this result compare to the vibronic coupling responsible for...
-
Among the naturally occurring minerals of Al are diaspore (-AlO(OH)), boehmite (-AlO(OH)) and gibbsite (-Al(OH) 3 ). At low pH, reactions with H + give rise to water-soluble [Al(OH 2 ) 6 ] 3+ ,...
-
(a) Comment why, in Fig. 13.1, the data are presented on a logarithmic scale. What are the relative abundances of Al (Fig. 13.1) and Mg (Fig. 12.2) in the Earths crust? (b) Show that the changes in...
-
Comprehensive problem-calculate missing amounts, issue price, net income, and dividends; interpret stock dividend and split. Bacon, Inc., has the following stockholders' equity section in its May 31,...
-
Rewrite Programming Exercise 7.5 using streams. Display the numbers in increasing order. Data from Programming Exercise 7.5 Write a program that reads in 10 numbers and displays the number of...
-
If variable manufacturing overhead is applied to production on the basis of direct labor- hours and the direct labor efficiency variance is unfavorable, will the variable overhead efficiency variance...
-
What is a statistical control chart, and how is it used?
-
Why can undue emphasis on labor efficiency variances lead to excess work in process inventories?
-
While a listing was in fierce, the broker sent a letter to the owner saying that she had obtained a $500.00 deposit on the purchase price of the property. She did not reveal the name of the buyer or...
-
The Calendar Company expects to have $4,000 in cash on hand at the beginning of January, and the company's target cash balance is $2,000. Net cash flow for January is minus $10,000. The company...
-
Taser Company owns the following assets acquired on January 1, 2020. Asset Cost Residual Value Estimated Useful Life A $48,000 $800 10 years B 12,000 - 5 years C 70,000 1,600 8 years a. Compute the...

Study smarter with the SolutionInn App


