Refer to the periodic table on the inside front cover, and arrange the following in the expected
Question:
Refer to the periodic table on the inside front cover, and arrange the following in the expected order of increasing first ionization energy: As, Sn, Br, Sr.
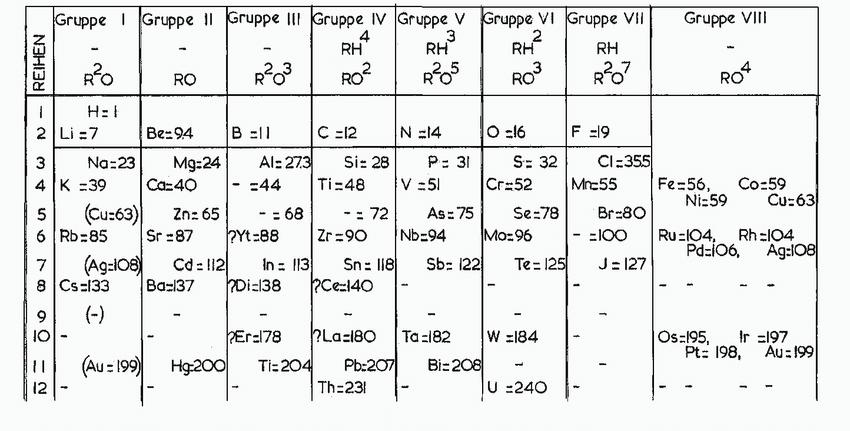
Transcribed Image Text:
REIHEN 3 A W 1 2 Li=7 4 K 56 94 Gruppe 1 Gruppe 11 Gruppe III Gruppe IV Gruppe V Gruppe VI Gruppe VII RH²³ 3 2.5 R²3 7 9 10 12 M 6 Rb 85 R²0 - H=1 - 8 Cs-133 (-) (Au=199) (Cu=63) Na:23 =39 Ca=40 (Ag=108) - RO Be=94 Mg:24 Cd: 112 B =11 Ba=137 - Al: 273 Zn: 65 Sr=87 ?Yt=88 =44 -=68 RH RO² Di=138 C =12 Si: 28 Ti=48 N=14 P= 31 V = 51 Hg:200 Ti=204 Pb:207 Th=231 -= 72 Zr=90 Nb=94 As: 75 In = 113 Sn: 118 Sb: 122 ?Ce=140 RH² 2 RO³ Bi-208 O=16 S= 32 Cr=52 Se=78 Mo=96 Er=178 ?La-180 Ta=182 W =184 Te = 125 L U =240 RH R²O7 F =19 CI=355 Mr-55 Br=80 - =100 J=127 Gruppe VIII Fe=56, RO4 Ni:59 Co=59 Cu:63 Ru-104, Rh=104 Pd=106, Ag:108 Os=195, Ir =197 Pt: 198, Au 199
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
Analyze Ionization energies decrease as atomic radii increase Thus if we arrange these four atoms ac...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) Refer to the periodic table on the inside front cover, and arrange the following in the expected order of increasing first ionization energy Cl, K, Mg, S. (B) Refer to the periodic table on the...
-
Refer only to the periodic table on the inside front cover, and arrange the following ionization energies in order of increasing value: the first ionization energy of F; the second ionization energy...
-
Refer only to the periodic table on the inside front cover and indicate which of the atoms, Bi, S, Ba, As, and Ca, (a) Is most metallic; (b) Is most nonmetallic; (c) Has the intermediate value when...
-
11. Which one is not a source for collecting references? Ans : O Proquest Ebscohost O Scopus Safe assign
-
a. Show the reconciling items in a horizontal model or write the adjusting journal entry (or entries) that should be prepared to reflect the reconciling items of Exercise 5.2. b. What is the amount...
-
How can the study of contemporary foragers provide us with an understanding of Paleolithic lifestyles?
-
p. 510 Is it possible to be a great leader of employees in a highly mechanistic organization? What special talents or abilities might be required?
-
On February 1, 2010, Buffalo Corporation issued 3,000 shares of its $5 par value common stock for land worth $31,000. Prepare the February 1, 2010, journal entry.
-
5 QUESTION 5 Luke Corporation produces a variety of products, each within their own division. Last year, the managers at Luke developed and began marketing a new chewing gum, Bubbs, to sel in vending...
-
Which of the following species has the greatest number of unpaired electrons (a) Ge; (b) Cl; (c) Cr 3+ ; (d) Br - ?
-
Which of the following species would you expect to be diamagnetic and which paramagnetic? (a) K + ; (b) Cr 3+ ; (c) Zn 2+ ; (d) Cd; (e) Co 3+ ; (f) Sn 2+ ; (g) Br.
-
Let p = (p 1 , . . . ,p n ) be a list of probabilities with p 1 + + p n = 1. Write a function coupon(n, p) which generalizes the function, above, and simulates the coupon collectors problem for...
-
Low Desert Pottery works makes a variety of pottery products that it sells to retailers. The company uses a job-order costing system in which departmental predetermined overhead rates are used to...
-
ASSESSMENT CPCCBC5002A Monitor costing systems on medium rise building and construction projects Please provide answer to Part 2 - Monitor expenditure for a medium-rise project as per below...
-
Questions 6-8 refer to the same problem A sinusoidal wave with wavelength 2 m and amplitude 5 mm is traveling along the x axis. The wave is traveling in the -x direction at a speed of 2m/s At t = Os,...
-
Consider a circuit where one or more capacitors is discharged through a light bulb filament with a resistance of 3.0 0.3 . Assume that the resistance of the filament is constant (to within the stated...
-
3. For a vibrating string of length with fixed ends, each mode of vibration can be written as where wk ux(x, t) = M* sin(wxt + k) sin(x) and Mk, Ok are determined by initial conditions. For all k >...
-
Consider two nodes, A and B, that use the slotted ALOHA protocol to contend for a channel. Suppose node A has more data to transmit than node B. and node A's retransmission probability PAis greater...
-
A liquid flows upward through a valve situated in a vertical pipe. Calculate the differential pressure (kPa) between points A and B. The mean velocity of the flow is 4.1 m/s. The specific gravity of...
-
Huron Company produces a commercial cleaning compound known as Zoom. The direct materials and direct labor standards for one unit of Zoom are given below: During the most recent month, the following...
-
Refer to the data in Exercise 118. Assume that instead of producing 4,000 units during the month, the company produced only 3,000 units, using 14,750 pounds of material. (The rest of the material...
-
Erie Company manufactures a small CD player called the Jogging Mate. The company uses standards to control its costs. The labor standards that have been set for one Jogging Mate CD player are as...
-
As a staff accountant at a company that provides life insurance, the controller has asked you to prepare a report on the assumptions related to settlement costs needed to calculate the liability for...
-
OMEGA Hotel provides a type of rooms with a sale price of 50 euros. Its total fixed cost amounts to 100,000 euros. The variable cost per room was estimated at 25 euros. The dead point in rooms is:...
-
You are required to use a financial calculator or spreadsheet (Excel) to solve 10 problems (provided on page 5) on the applications of the time value of money. You are required to show the following...

Study smarter with the SolutionInn App


